Introduction & Epidemiology
Bone matrix forms the foundational non-cellular component of bone tissue, providing its characteristic strength, rigidity, and metabolic reservoir functions. Understanding its intricate composition and dynamic properties is paramount for orthopedic surgeons, as nearly all interventions, from fracture management to arthroplasty, rely on the successful interaction with, or regeneration of, this complex biological material. Bone matrix is a dynamic composite material, constantly undergoing remodeling orchestrated by osteoblasts, osteocytes, and osteoclasts, a process crucial for maintaining skeletal integrity and adapting to mechanical demands. Disruptions in matrix homeostasis underpin a wide array of orthopedic pathologies, including non-unions, osteomalacia, osteoporosis, and the failure of orthopedic implants.
The dry weight of bone matrix is predominantly composed of two principal components: organic and inorganic constituents, present in approximately 40% and 60% ratios, respectively. This precise balance is critical for providing both the flexibility and tensile strength characteristic of the organic phase, and the compressive strength and hardness imparted by the inorganic mineral phase. Epidemiologically, conditions affecting bone matrix are widespread. Osteoporosis, characterized by decreased bone mineral density and microarchitectural deterioration of bone tissue, affects millions globally, leading to fragility fractures. Non-union rates after long bone fractures range from 5-10%, often due to inadequate matrix formation or integration. The success of arthrodesis procedures and the longevity of uncemented arthroplasty components are directly dependent on robust bone matrix regeneration and osseointegration. Therefore, a comprehensive understanding of bone matrix biology is fundamental for effective orthopedic practice.
Organic Components of Bone Matrix
The organic matrix constitutes approximately 40% of the dry weight of bone. Its primary role is to provide tensile strength and resilience, preventing brittle fracture.
-
Collagen: Type I collagen is the most abundant protein in the body, accounting for over 90% of the organic components of bone matrix. It is synthesized by osteoblasts and arranged in a highly organized manner.
- Predominantly Type I Collagen: The predominance of type I collagen (mnemonic: bone contains the word one ) is critical for the tensile strength of bone. It forms a hierarchical structure, beginning with tropocollagen molecules that spontaneously assemble into microfibrils, which then aggregate into larger collagen fibrils. These fibrils are further organized into collagen fibers and lamellae.
-
Structural Features:
- Hole zones (gaps): These are discrete regions within the collagen fibril, existing between the ends of adjacent tropocollagen molecules within the stagger.
- Pores: These are found between the sides of parallel collagen molecules.
- Mineral Deposition: Crucially, these hole zones and pores serve as the primary sites for initial mineral deposition (calcification) in the form of hydroxyapatite crystals, linking the organic framework to the inorganic component.
- Cross-linking: Collagen molecules undergo extensive enzymatic cross-linking, predominantly by lysyl oxidase. This process decreases collagen solubility and significantly increases its tensile strength, conferring the ability to resist significant pulling forces without yielding. Abnormalities in collagen cross-linking, as seen in conditions like osteogenesis imperfecta or Ehlers-Danlos syndrome, lead to compromised bone integrity.
-

-

-
Proteoglycans: These are macromolecules consisting of a protein core with covalently attached glycosaminoglycan (GAG) chains (e.g., chondroitin sulfate, keratan sulfate). In bone matrix, proteoglycans like decorin, biglycan, and fibromodulin play multiple roles:
- They regulate collagen fibril formation and organization.
- They bind growth factors (e.g., BMPs, TGF-β), influencing their availability and activity within the matrix.
- They contribute to the viscoelastic properties of bone.
- They are involved in mineralization processes, potentially inhibiting or promoting crystal growth depending on their specific structure and location.
-
Matrix Proteins (Noncollagenous): These constitute a smaller but functionally critical fraction of the organic matrix, typically 5-10% of the total protein. They regulate bone formation, resorption, and mineralization.
-
Osteocalcin:
This is the most abundant noncollagenous protein in bone and is exclusively produced by osteoblasts.
- It is a vitamin K-dependent protein that binds strongly to hydroxyapatite, suggesting a role in mineralization and crystal maturation.
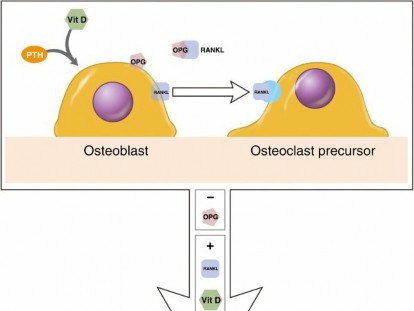
- Its synthesis is inhibited by parathyroid hormone (PTH) and stimulated by 1,25(OH)2D3 (calcitriol).
- Due to its specific production by osteoblasts and release into circulation, osteocalcin can be measured in serum or urine as a biochemical marker of bone turnover, reflecting osteoblast activity.
- Osteonectin (SPARC - Secreted Protein Acidic and Rich in Cysteine): Plays a role in cell-matrix interactions, binding to collagen, hydroxyapatite, and various growth factors. It is involved in regulating mineralization and promoting cell attachment.
- Osteopontin (OPN): A glycoprotein with RGD (arginine-glycine-aspartate) sequences that mediate cell adhesion via integrin receptors. It is involved in mineral formation and dissolution, acting as both an inhibitor and promoter of mineralization. OPN is also implicated in osteoclast attachment to the bone surface.
- Bone Sialoprotein (BSP): Strongly expressed by osteoblasts during mineralization, BSP binds to collagen and hydroxyapatite. It is thought to nucleate hydroxyapatite crystals, initiating mineralization.
- Fibronectin: A large glycoprotein involved in cell adhesion, migration, and differentiation. It binds to collagen, proteoglycans, and cell surface receptors, facilitating cell-matrix interactions during bone remodeling and repair.
- Growth Factors and Cytokines: The bone matrix serves as a reservoir for numerous growth factors and cytokines, including bone morphogenetic proteins (BMPs), transforming growth factor-beta (TGF-β), insulin-like growth factors (IGFs), and fibroblast growth factors (FGFs). These factors are sequestered within the matrix and released during resorption, influencing local cell activity and orchestrating bone formation and repair. For example, some factors like IL-10 have been shown to suppress osteoclasts, which are critical for bone remodeling.
-
Osteocalcin:
This is the most abundant noncollagenous protein in bone and is exclusively produced by osteoblasts.
Inorganic (Mineral) Components of Bone Matrix
The inorganic component constitutes approximately 60% of the dry weight of bone and is primarily responsible for its hardness and compressive strength.
-
Hydroxyapatite:
The principal mineral in bone is a crystalline form of calcium phosphate, specifically hydroxyapatite [Ca10(PO4)6(OH)2].
- These crystals are exquisitely small and arranged in close apposition to the collagen fibrils, particularly within the hole zones and pores, forming a nanostructured composite material.
- The intimate relationship between collagen and hydroxyapatite is crucial for bone's biomechanical properties, allowing it to resist both tensile and compressive forces effectively.
- The mineral phase also acts as a vast reservoir for calcium, phosphate, and other ions, making bone a critical regulator of systemic mineral homeostasis.
- Other Ions: In addition to calcium and phosphate, the mineral phase incorporates trace amounts of other ions, including magnesium, strontium, carbonate, citrate, and fluoride, which can influence crystal structure and bone quality.
Surgical Anatomy & Biomechanics
The anatomical organization of bone matrix dictates its biomechanical properties and how it responds to mechanical loading and surgical intervention. Bone exists in two main forms: cortical (compact) bone and cancellous (trabecular) bone, each with distinct matrix architecture and biomechanical profiles.
Cortical Bone Matrix
Cortical bone forms the dense outer shell of most bones, comprising approximately 80% of skeletal mass. Its matrix is highly organized into osteons (Haversian systems), which are concentric lamellae of bone matrix surrounding a central Haversian canal containing blood vessels and nerves.
- Lamellar Structure: The collagen fibers within each lamella are arranged in a highly ordered, often helical pattern, with the orientation changing between adjacent lamellae. This plywood-like arrangement provides exceptional resistance to torsional forces and crack propagation.
- Interstitial Lamellae: These are remnants of old osteons, contributing to the overall density and strength of cortical bone.
- Perforating (Volkmann's) Canals: These canals connect adjacent Haversian canals and the periosteal and endosteal surfaces, facilitating nutrient and waste exchange throughout the dense matrix.
- Osteocytes: Osteocytes, terminally differentiated osteoblasts, reside within lacunae embedded within the matrix. They extend dendritic processes through canaliculi, forming a vast lacuno-canalicular network that allows for mechanosensing and communication, regulating matrix remodeling in response to mechanical stimuli.
Cancellous Bone Matrix
Cancellous bone, found within the ends of long bones, vertebral bodies, and flat bones, forms a porous, honeycomb-like network of trabeculae. Despite its lower density, it is metabolically highly active and crucial for energy absorption and load distribution.
- Trabecular Architecture: The matrix in cancellous bone is organized into slender, interconnected trabeculae. The orientation of these trabeculae is not random but adapts to the dominant stress lines, optimizing the bone's ability to resist compressive and shear forces (Wolff's Law).
- High Surface Area: The porous nature of cancellous bone provides a large surface area for metabolic activity and interaction with bone marrow elements. This high surface area also makes it a rich source of osteoprogenitor cells and growth factors, critical for bone healing.
- Mechanical Properties: Cancellous bone exhibits anisotropic properties, meaning its mechanical strength varies with the direction of applied load. It is highly deformable and capable of absorbing significant energy before fracture, due to its ability to undergo microfractures and plastic deformation without catastrophic failure.
- Remodeling: The high turnover rate of cancellous bone matrix makes it particularly susceptible to metabolic disturbances (e.g., osteoporosis), leading to trabecular thinning, perforation, and loss of connectivity, severely compromising its mechanical integrity.
Biomechanical Significance of Bone Matrix Components
The unique composite nature of bone matrix, with its organic and inorganic phases, confers its remarkable biomechanical properties:
- Tensile Strength: Primarily provided by the type I collagen network and its extensive cross-linking. Collagen's fibrillar structure and its ability to absorb energy contribute significantly to the bone's resistance to pulling forces.
- Compressive Strength: Largely attributed to the inorganic hydroxyapatite crystals. These stiff mineral crystals resist shortening under load, distributing forces effectively.
- Flexibility and Toughness: The interaction between collagen and mineral provides bone with a degree of flexibility and toughness, preventing brittle fracture. The organization of collagen fibrils and mineral crystals at multiple hierarchical levels allows for energy dissipation and crack deflection.
- Viscoelasticity: Bone exhibits viscoelastic properties, meaning its response to load is dependent on the rate of loading. This is partly due to the fluid within the lacuno-canalicular network and the inherent properties of the organic matrix components.
- Fatigue Resistance: The ability of bone matrix to undergo continuous remodeling is essential for repairing microdamage that accumulates with repetitive loading, thus preventing catastrophic fatigue failure. Osteocyte mechanotransduction plays a key role in orchestrating this adaptive remodeling.
Understanding these structural and biomechanical principles is critical for surgical planning, including implant design, bone graft selection, and optimizing fixation strategies. For instance, the greater stiffness of cortical bone makes it suitable for structural grafts, while the rich cellularity and surface area of cancellous bone make it superior for osteoinduction and osteoconduction in reconstructive procedures.

Indications & Contraindications
The indications and contraindications for orthopedic interventions, when viewed through the lens of bone matrix, fundamentally revolve around situations where the intrinsic quality, quantity, or regenerative capacity of the bone matrix is either compromised or requires enhancement. This perspective guides decisions on bone grafting, the use of biologics, and strategies for managing a myriad of skeletal pathologies.
Indications for Interventions Targeting Bone Matrix
Interventions are typically indicated when the native bone matrix is insufficient for structural support, requires augmentation for healing, or needs modification to facilitate integration.
-
Non-unions and Delayed Unions:
The failure of a fracture to heal within an expected timeframe, often due to inadequate bone matrix formation, poor vascularity, or mechanical instability.
- Operative: Bone grafting (autograft, allograft), biologic augmentation (BMPs, PRP), internal or external fixation to provide stability and promote matrix deposition.
-
Critical Size Defects (CSDs):
Bone defects that are too large to heal spontaneously by intrinsic bone regeneration. These require the introduction of new matrix material.
- Operative: Large volume autograft or allograft, vascularized bone transfers, custom implants, or scaffolds combined with biologics.
-
Arthrodesis (Fusion Procedures):
Intentional fusion of a joint requires robust bone matrix bridging across the joint space.
- Operative: Decortication of joint surfaces, bone grafting (typically autograft or DBM), internal fixation to provide stability.
-
Spinal Fusion:
Similar to joint arthrodesis, fusion of vertebral segments relies on the formation of a solid bony bridge.
- Operative: Interbody fusion devices with bone graft, posterolateral fusion with autograft/allograft/biologics, instrumentation.
-
Osteochondral Defects:
Focal cartilage and subchondral bone loss, where the subchondral bone matrix is compromised.
- Operative: Osteochondral autograft transfer (OATS), allograft transplantation, matrix-induced autologous chondrocyte implantation (MACI), microfracture to stimulate fibrocartilaginous matrix repair.
-
Augmentation of Impaired Host Bone:
In cases of severe osteoporosis, metabolic bone disease, or revision arthroplasty with significant bone loss, existing bone matrix may be insufficient to support implants.
- Operative: Use of structural bone grafts, cement augmentation, adjunctive pharmacotherapy to improve host bone quality.
-
Osseointegration for Arthroplasty/Trauma Implants:
Ensuring optimal bone matrix growth onto and into implant surfaces for long-term stability.
- Operative: Surface treatments of implants (porous coatings, hydroxyapatite coatings), specific implant designs (press-fit), potential use of biologics in challenging cases.
-
Treatment of Metabolic Bone Diseases (Adjunctive):
While primarily managed medically, surgical intervention may be needed for resultant fractures or deformities, requiring a thorough understanding of the compromised matrix.
- Operative: Fixation of fragility fractures, osteotomies for deformity correction, often with consideration for bone quality.
Contraindications for Interventions Targeting Bone Matrix
Contraindications often relate to host factors that preclude successful matrix regeneration or integration, or situations where the risks outweigh the benefits.
- Active Infection: Infection at the surgical site is an absolute contraindication for introducing bone grafts or implants, as it severely impairs matrix regeneration and promotes graft/implant failure.
- Severe Systemic Illness/Poor Nutritional Status: Conditions that compromise the patient's overall health and metabolic capacity for healing (e.g., uncontrolled diabetes, severe malnutrition, chronic renal failure) can lead to poor matrix formation and wound healing complications.
- Inadequate Vascularity: Insufficient blood supply to the recipient site significantly impedes bone matrix formation and remodeling.
- Uncontrolled Metabolic Bone Disease: Untreated osteoporosis, osteomalacia, or other metabolic bone disorders can lead to poor bone quality and high failure rates for interventions.
- Poor Patient Compliance: Non-compliance with post-operative weight-bearing restrictions or rehabilitation protocols can disrupt the healing matrix, leading to failure.
- Irradiated Bone: Prior radiation therapy can significantly compromise the vascularity and cellularity of bone, impairing matrix healing.
- Excessive Mechanical Instability: While addressing instability is often an indication, excessive, uncontrolled motion at a fracture or fusion site will prevent organized bone matrix formation, leading to fibrous tissue instead.
- Limited Bone Stock (Donor Site): For autograft procedures, insufficient donor bone stock can be a relative contraindication, necessitating the use of allograft or synthetic alternatives.
- Hypersensitivity/Allergy: Rare, but potential contraindication to specific graft or biologic components.
Table: Operative vs. Non-Operative Indications Related to Bone Matrix
| Indication Category | Operative Management (Direct Matrix Manipulation/Augmentation) | Non-Operative Management (Indirect Matrix Support/Optimization) |
|---|---|---|
| Fracture Healing | Bone grafting (autograft/allograft) for non-unions, critical size defects. Internal/external fixation. Biologic adjuncts (BMPs). | Immobilization (casting/bracing). Pain management. Nutritional optimization (Vit D, Ca). Smoking cessation. Fracture liaison. |
| Arthrodesis/Fusion | Decortication, autograft/allograft, interbody cages, instrumentation for stability, biologic adjuncts. | Spinal orthoses (post-op). Specific activity restrictions (post-op). Pain management. |
| Bone Loss/Defects | Structural bone grafts, vascularized bone transfers, synthetic scaffolds, cement augmentation (e.g., vertebroplasty). | Nutritional support, pharmacotherapy for underlying osteoporosis/osteomalacia. |
| Implant Osseointegration | Porous/hydroxyapatite coated implants, specific surface treatments, potentially local biologics. | Careful patient selection (bone quality assessment), appropriate implant sizing/preparation. |
| Osteochondral Repair | OATS, allograft, MACI, microfracture, particulate cartilage grafting. | Activity modification, physical therapy, bracing, NSAIDs. |
| Metabolic Bone Disease (Secondary) | Fixation of pathological fractures, deformity correction osteotomies, revision arthroplasty. | Pharmacotherapy (bisphosphonates, PTH analogs, denosumab), Vitamin D/Calcium supplementation, fall prevention. |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning for interventions involving bone matrix is crucial, extending beyond standard surgical preparations to specifically address the biological milieu and mechanical environment conducive to successful matrix regeneration and integration.
Pre-Operative Planning
-
Comprehensive Patient Assessment:
- Medical Comorbidities: Identify and optimize systemic conditions that impair bone healing (e.g., diabetes, renal insufficiency, malnutrition, vascular disease, autoimmune disorders). Consultation with internal medicine specialists may be warranted.
- Medication Review: Discontinue or adjust medications known to inhibit bone healing (e.g., NSAIDs, corticosteroids, tobacco products). Discuss anti-resorptive agents with endocrinology/rheumatology.
- Nutritional Status: Assess vitamin D levels, calcium intake, and overall nutritional state. Supplementation should be initiated pre-operatively if deficiencies are identified.
- Smoking Cessation: Aggressive counseling and cessation programs are critical due to the profound negative impact of nicotine on vascularity and osteogenesis.
-
Detailed Imaging Review:
- Radiographs: Assess fracture patterns, bone defects, pre-existing deformities, and overall bone quality.
- CT Scans: Provide critical 3D information on defect size, bone stock, precise non-union morphology, and cortical integrity, especially in complex cases (e.g., pelvic or vertebral defects, critical size defects in long bones). Allow for planning of graft size and morphology.
- MRI: Useful for assessing soft tissue envelopes, vascularity, and differentiating fibrous non-union from viable bone.
- Bone Scans/PET Scans: Can help assess metabolic activity at a non-union site or graft incorporation.
-
Bone Grafting Strategy:
- Autograft Selection: Plan the donor site (e.g., iliac crest, proximal tibia, distal femur, reamer-irrigator-aspirator (RIA) system) based on the required volume, structural demands, and cellularity. Considerations include patient morbidity, surgical access, and bone quality at the donor site.
-
Allograft Selection:
Determine the type of allograft (cancellous, cortical, DBM, structural) based on the specific needs:
- Cancellous: Osteoconductive and osteoinductive, good for filling voids.
- Cortical: Structural support.
- DBM: Osteoinductive properties.
- Structural: For large segmental defects.
- Synthetic Grafts/Biologics: Consider their role as extenders or standalone options, especially for less critical defects or in conjunction with autograft. Plan for their proper preparation and application.
- Volume Estimation: Accurately estimate the volume of graft material required to fill defects or achieve fusion.
-
Implant Selection and Fixation Strategy:
- Choose implants (plates, screws, rods, cages) that provide optimal mechanical stability for the intended healing environment, considering the quality of the surrounding bone matrix.
- Plan for appropriate screw length and diameter, and consider locking plate technology for osteopenic bone.
- For arthroplasty, plan for cemented vs. uncemented components based on bone stock and patient factors.
- Contingency Planning: Develop alternative strategies for unexpected findings, such as inadequate bone stock, poor vascularity, or intraoperative complications.
Patient Positioning
Patient positioning must facilitate optimal surgical exposure of both the recipient site and any planned bone graft donor sites, while ensuring patient safety and comfort.
- Recipient Site Exposure: The specific surgical approach for the primary pathology dictates the basic positioning (e.g., supine for anterior hip, lateral for femur, prone for posterior spine).
-
Donor Site Access:
- Iliac Crest: If posterior iliac crest harvest is planned, a lateral decubitus or prone position is common. For anterior crest, supine with slight hip flexion and external rotation may be used.
- Proximal Tibia: Supine position with knee slightly flexed.
- Distal Femur (RIA): Supine or lateral, ensuring the reaming trajectory is clear.
- Radiographic Control: Ensure unrestricted access for intraoperative fluoroscopy or conventional radiography for accurate graft placement and implant fixation.
- Pressure Point Padding: Meticulous padding of all pressure points is essential to prevent nerve palsies or skin breakdown during prolonged procedures.
- Hemostasis: Positioning may need to optimize venous return or reduce blood flow to the surgical field (e.g., slight Trendelenburg for lower extremity, controlled hypotension).
- Tourniquet Placement: If a tourniquet is used for extremity surgery, ensure proper placement and padding.
Detailed Surgical Approach / Technique
The surgical approach and technique for manipulating bone matrix are highly dependent on the specific clinical scenario, but generally involve principles of debridement, optimization of the biological environment, introduction of osteoinductive/osteoconductive materials, and mechanical stabilization. This section focuses on generalized strategies for enhancing bone matrix healing and regeneration.
General Principles of Bone Matrix Enhancement
-
Debridement and Preparation of Recipient Site:
- Removal of Non-viable Tissue: Resect all necrotic or avascular bone and soft tissue. This creates a healthy environment for vascular ingrowth and new matrix formation.
- Decortication: For non-unions or fusion sites, extensive decortication of cortical bone is performed to expose cancellous bone and its rich blood supply. This stimulates a local inflammatory response and provides a larger surface area for osteogenic cell migration and matrix deposition.
- Meticulous Hemostasis: Ensure a dry field. Excessive bleeding can create a hematoma that impedes revascularization and matrix integration.
-
Optimizing the Biological Environment:
- Local Growth Factor Delivery: The addition of bone morphogenetic proteins (BMPs) can directly induce mesenchymal stem cell differentiation into osteoblasts, promoting new bone matrix formation. BMP-2 and BMP-7 are commonly used.
- Cellular Augmentation: Autologous sources of osteoprogenitor cells, such as bone marrow aspirate concentrate (BMAC) or mesenchymal stem cells (MSCs) delivered with a scaffold, can enhance local osteogenic potential.
- Platelet-Rich Plasma (PRP): While controversial, PRP contains numerous growth factors that may theoretically enhance local healing, though evidence in bone matrix regeneration is mixed.
-
Introduction of Matrix-Providing Materials (Bone Grafts):
-
Autograft:
The gold standard, possessing osteoconductive, osteoinductive, and osteogenic properties.
-
Cancellous Autograft:
Harvested from sites like the iliac crest or proximal tibia. Ideal for filling defects, providing osteoprogenitor cells and growth factors. It provides a scaffold for ingrowth and is eventually remodeled.
- Technique: Typically harvested using curved osteotomes or curettes. Care must be taken to maintain the outer cortical integrity of the donor site to minimize fracture risk.
-
Cortical Autograft:
Harvested from sites like the fibula or ribs (vascularized) or iliac crest (non-vascularized). Provides structural support but has limited osteogenic potential.
- Technique: Requires careful dissection to preserve neurovascular structures if vascularized.
-
Reamer-Irrigator-Aspirator (RIA) System:
Provides a large volume of high-quality cancellous bone and bone marrow aspirate from the femoral or tibial canal, with potentially reduced donor site morbidity compared to iliac crest.
- Technique: Reaming is performed under irrigation, and the aspirate is collected in a specialized trap.
-
Cancellous Autograft:
Harvested from sites like the iliac crest or proximal tibia. Ideal for filling defects, providing osteoprogenitor cells and growth factors. It provides a scaffold for ingrowth and is eventually remodeled.
-
Allograft:
Bone from a cadaveric donor. Osteoconductive, can be osteoinductive (demineralized bone matrix, DBM) but lacks osteogenic cells.
- Cancellous Allograft: Used for filling defects, similar to cancellous autograft. Less osteogenic but provides osteoconductive scaffold.
- Cortical Allograft: Provides structural support (e.g., segmental defects, reconstruction after tumor resection). Incorporates more slowly than autograft and has potential for immunogenicity and non-union.
- Demineralized Bone Matrix (DBM): Processed allograft with preserved collagen and non-collagenous proteins, including growth factors. Primarily osteoinductive, with variable osteoconductivity. Available in various forms (gel, putty, chips).
-
Synthetic Bone Grafts:
Osteoconductive scaffolds, often calcium phosphate-based (e.g., hydroxyapatite, beta-tricalcium phosphate) or bioglass. Lack osteoinductive and osteogenic properties. Used as extenders or for non-critical defects.
- Technique: Applied directly to the defect site. May be combined with BMAC or biologics to enhance osteoinductivity.
-
Autograft:
The gold standard, possessing osteoconductive, osteoinductive, and osteogenic properties.
-
Mechanical Stability (Fixation):
- Rigid Fixation: Essential for successful bone matrix formation and remodeling. Movement at the fracture or fusion site beyond a critical threshold promotes fibrous tissue formation rather than bone (Pauwels' classification).
- Implants: Plates, screws, intramedullary nails, external fixators, interbody cages, and other devices are chosen to provide appropriate stability.
- Compression: In some scenarios, controlled compression across the healing site can enhance bone healing by increasing interfragmentary strain within optimal ranges.
Step-by-Step Approach for a Non-Union with Bone Defect (Example)
Consider a challenging long bone non-union with a significant bone defect, requiring bone matrix augmentation.
-
Pre-Operative Planning:
- Thorough clinical assessment, identify and address comorbidities.
- Advanced imaging (CT) to define defect size, non-union characteristics, and plan fixation.
- Select bone graft source and type (e.g., large volume autograft from RIA or iliac crest, potentially supplemented with DBM or BMP).
- Plan definitive fixation strategy (e.g., revision intramedullary nail, locking plate).
-
Patient Positioning:
- Typically supine or lateral decubitus, depending on the affected bone and planned donor site.
- Ensure appropriate padding, tourniquet application if needed, and fluoroscopy access.
-
Surgical Approach:
- Exposure of Non-Union Site: Utilize a standard approach to expose the involved bone segment. Careful dissection to minimize further soft tissue damage and preserve any viable periosteum.
- Debridement and Resection: Identify and resect all fibrous tissue, avascular bone, and sclerotic margins from the non-union site. This creates fresh, bleeding bone ends.
- Decortication: Aggressively decorticate the cortical bone surrounding the non-union, exposing cancellous bone and stimulating local blood supply. This is crucial for creating a biologically active environment for matrix ingrowth.
- Correction of Deformity (if present): Achieve anatomical alignment and length.
- Medullary Canal Preparation: If an intramedullary nail is used, ream the canal to remove fibrous tissue and expose healthy marrow, allowing for the passage of a larger diameter nail.
-
Bone Graft Harvest (Simultaneous or Sequential):
- If RIA, perform femoral or tibial reaming and aspiration.
- If iliac crest, make a separate incision over the posterior or anterior iliac crest. Use osteotomes and curettes to harvest cancellous bone, packing it into a sterile container. Avoid cortical perforation to prevent pelvic instability.
-
Graft Placement and Compaction:
- Pack the cancellous autograft directly into the defect and around the non-union site, ensuring intimate contact with the decorticated host bone. The graft provides osteoconductive scaffold and osteoinductive/osteogenic factors.
- If using DBM or synthetic grafts, mix with autograft as an extender or place as per manufacturer instructions.
- Consider local application of BMPs or BMAC directly into the graft site, mixed with the graft material or placed as a gel.
- Definitive Fixation: Apply the chosen implant (e.g., revision IM nail, locking plate) to provide absolute or relative stability. The construct must be rigid enough to prevent excessive motion, which would disrupt early matrix formation. Achieve robust screw purchase in healthy bone proximal and distal to the non-union.
- Wound Closure: Layered closure with attention to soft tissue coverage and tension. Drains may be used to manage hematoma.
Internervous Planes
While not explicitly about bone matrix, respecting internervous planes during dissection is critical for minimizing morbidity, preserving function, and optimizing patient recovery, which indirectly contributes to the overall success of the healing process by reducing pain and enhancing rehabilitation. For example:
- Proximal Femoral Fractures: Lateral approach through the tensor fascia lata avoids major nerves.
- Tibial Fractures: Anterolateral approach between tibialis anterior and extensor digitorum longus, or direct medial approach to avoid anterior compartment structures.
- Spinal Surgery: Minimally invasive techniques aim to minimize muscle and nerve retraction, preserving the vascularity and cellular integrity of the surrounding tissues, which in turn supports bone matrix healing.
Complications & Management
Complications related to bone matrix interventions are diverse, ranging from issues directly related to graft incorporation to general surgical risks. Understanding these complications and their management is crucial for optimal patient outcomes.
Complications Related to Bone Matrix Interventions
-
Non-Union/Mal-Union:
- Incidence: Varies significantly with fracture type, location, patient factors, and surgical technique (5-10% for long bones, higher for specific sites like scaphoid or navicular).
- Etiology: Inadequate vascularity, insufficient biological activity (lack of osteoprogenitor cells/growth factors), mechanical instability (excessive motion), infection, poor nutrition, smoking. Essentially, a failure of new bone matrix to bridge the defect.
-
Management:
- Biological: Bone grafting (autograft is preferred), biologic augmentation (BMPs, BMAC).
- Mechanical: Revision fixation with more rigid construct (e.g., plate and screws, IM nail), external fixator.
- Systemic: Optimization of nutrition, vitamin D, smoking cessation, treatment of underlying metabolic bone disease.
- Debridement: Resection of intervening fibrous tissue and sclerotic bone, decortication.
-
Graft Resorption/Collapse:
- Incidence: Higher for allografts, especially structural allografts, due to slower incorporation and lack of viable cells. Can occur with autograft if mechanically overloaded or poorly vascularized.
- Etiology: Avascularity, infection, excessive mechanical loading leading to fatigue failure, immunologic reaction (allograft). The host bone fails to remodel and integrate the graft matrix effectively.
-
Management:
- Early: Protected weight-bearing, bracing.
- Significant Resorption/Failure: Revision surgery with re-grafting (often autograft) and more rigid fixation.
- Infection: Debridement, antibiotics, potential graft removal.
-
Infection:
- Incidence: 1-5% for elective orthopedic surgery, higher for open fractures or revision surgeries.
- Etiology: Bacterial contamination during surgery or post-operatively. Directly impairs bone matrix formation and promotes destruction.
-
Management:
- Acute: Aggressive surgical debridement (irrigation and debridement, I&D), culture-specific intravenous antibiotics, possible implant retention if stable and adequately debrided.
- Chronic: Staged surgery (implant removal, extensive debridement, antibiotic spacer, then delayed reconstruction/re-implantation with new bone matrix material). May require local or free tissue transfer for soft tissue coverage.
-
Donor Site Morbidity (for Autograft):
- Incidence: Highly variable depending on harvest site and technique (e.g., iliac crest: 10-30%, including pain, sensory nerve injury, fracture). RIA lower overall morbidity.
- Etiology: Direct surgical trauma to nerves, blood vessels, periosteum; stress concentrators from bone harvest; poor wound closure.
-
Management:
- Pain: Analgesia, local anesthetic blocks, physical therapy.
- Nerve Injury: Conservative management (most resolve), rarely surgical exploration.
- Fracture: Activity modification, sometimes internal fixation for significant fractures. Prevention is key (conservative harvest, appropriate technique).
-
Vascular Injury/Nerve Damage:
- Incidence: Rare but potentially devastating.
- Etiology: Direct trauma during dissection, retraction, or hardware placement.
- Management: Immediate repair of vascular injury (consult vascular surgeon). Nerve repair if indicated, otherwise conservative management with close observation.
-
Allograft-Related Complications:
- Disease Transmission: Extremely rare with modern screening and processing (e.g., HIV, hepatitis C), but a theoretical risk.
- Immunogenicity: Can contribute to slower incorporation or graft failure, although less pronounced with demineralized/freeze-dried allografts compared to fresh allografts.
- Management: Adherence to strict tissue banking protocols. For immunogenicity, often managed as graft failure.
-
Complex Regional Pain Syndrome (CRPS):
- Incidence: 1-5% after extremity trauma or surgery.
- Etiology: Multifactorial, involving neurologic and inflammatory components, can be triggered by surgical trauma and prolonged pain.
- Management: Early recognition, multidisciplinary approach (physical therapy, pain management specialists, pharmacotherapy, nerve blocks).
Table: Common Complications, Incidence, and Salvage Strategies
| Complication | Incidence (%) | Salvage Strategy |
|---|---|---|
| Non-Union/Delayed Union | 5-10% (Long Bones), Higher (Scaphoid, Navicular) | Revision surgery: Debridement, decortication, biological augmentation (autograft, DBM, BMPs), revision fixation (more rigid implant, additional hardware), external fixator. Systemic optimization (nutrition, smoking cessation). |
| Graft Resorption/Collapse | 5-20% (Structural Allografts), Lower (Autograft) | Protected weight-bearing, bracing. Revision surgery: Re-grafting (often autograft), more rigid fixation. For infection, graft removal and staged reconstruction. |
| Surgical Site Infection (SSI) | 1-5% (Elective), Higher (Open Fractures, Revisions) | Acute: I&D, culture-specific IV antibiotics, potentially implant retention. Chronic: Staged surgery (implant removal, extensive debridement, antibiotic spacer), delayed reconstruction, soft tissue coverage. |
| Donor Site Morbidity (Autograft) | 10-30% (Iliac Crest), Lower (RIA) | Pain: Analgesia, local blocks, physical therapy. Nerve injury: Conservative management, rarely surgical exploration. Fracture: Activity modification, internal fixation if severe. |
| Vascular/Nerve Injury | <1% | Immediate repair of vascular injury (vascular surgeon consult). Nerve repair if indicated (neurosurgery/plastic surgery consult), otherwise conservative management. |
| Allograft Disease Transmission | Extremely Rare (<1 in 1.6 million) | Not a surgical salvage. Prevention by strict tissue banking. Managed as disease-specific pathology if transmission occurs. |
| Complex Regional Pain Syndrome | 1-5% (Extremity Trauma/Surgery) | Multidisciplinary pain management, physical therapy, pharmacotherapy (gabapentin, tricyclics), sympathetic blocks. Early recognition is key. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is critical for optimizing the integration and remodeling of the newly formed or grafted bone matrix, minimizing complications, and restoring function. Protocols must be tailored to the specific surgical procedure, the stability of the fixation, and the quality of the host bone matrix. The primary goals are to allow for biological healing while gradually restoring mechanical loading to promote beneficial matrix adaptation.
General Principles
- Protect the Healing Environment: The initial phase focuses on protecting the healing bone matrix from excessive stresses that could lead to disruption, non-union, or graft failure. This involves periods of immobilization or protected weight-bearing.
- Gradual Load Progression: Controlled, progressive mechanical loading is essential. Wolff's Law dictates that bone matrix remodels in response to mechanical stresses. Appropriate loading stimulates osteocyte activity, promoting osteogenesis, improving bone density, and enhancing the structural integrity of the healing matrix.
- Restore Range of Motion (ROM) and Strength: Early, controlled motion, where appropriate, can prevent joint stiffness and adhesions, while muscle strengthening stabilizes the operated segment and facilitates return to function.
- Pain and Edema Management: Effective control of pain and swelling is crucial for patient comfort and participation in therapy.
Phases of Rehabilitation (General Framework)
Phase I: Protection and Early Mobilization (Typically Weeks 0-6/8)
- Goals: Protect surgical site/graft, control pain/edema, maintain integrity of non-operated joints, initiate gentle muscle activation.
- Weight-Bearing (WB): Non-weight bearing (NWB) or touch-down weight-bearing (TDWB) is typical for lower extremity procedures involving significant bone matrix reconstruction (e.g., non-union repair, large allograft). Upper extremity may allow protected active/passive ROM.
- Immobilization: Bracing, casting, or splinting may be used to provide external support and limit motion, especially in cases of compromised fixation or poor bone quality.
- Range of Motion: Gentle active-assisted or passive ROM of adjacent joints (if not directly involved in the surgery) to prevent stiffness. For the operated joint, ROM may be severely restricted initially.
- Exercises: Isometric exercises for muscles around the operated site (without stressing the bone matrix), gait training with assistive devices, core stabilization.
- Patient Education: Crucial for adherence to weight-bearing restrictions, wound care, and recognizing signs of complications.
Phase II: Controlled Progressive Loading and Intermediate Mobilization (Typically Weeks 6/8-12/16)
- Goals: Gradually increase mechanical stress on healing bone matrix, improve ROM, restore muscle strength, progress functional activities.
- Weight-Bearing: Progress from TDWB to partial weight-bearing (PWB) and then to weight-bearing as tolerated (WBAT), based on radiographic evidence of bone matrix consolidation and clinical stability. Fluoroscopic assessment or serial radiographs are essential.
- Range of Motion: Increase active and active-assisted ROM, working towards full joint mobility. Address any soft tissue restrictions.
- Exercises: Progressive resistance exercises for strengthening, balance and proprioception training, initiation of low-impact cardiovascular activities.
- Manual Therapy: Soft tissue mobilization, scar management.
- Emphasis: The mechanical stimulus from increasing weight-bearing and muscle activity is critical at this stage for stimulating bone matrix remodeling and strengthening.
Phase III: Advanced Loading and Functional Integration (Typically Weeks 12/16 - 6+ Months)
- Goals: Restore full strength, power, endurance, agility, and return to sport or occupational activities. Further enhance bone matrix density and structural integrity.
- Weight-Bearing: Full weight-bearing without assistive devices.
- Range of Motion: Full, pain-free ROM in all planes.
- Exercises: High-level strengthening, plyometrics, sport-specific drills, impact loading activities (running, jumping) as appropriate, with strict monitoring for pain or adverse reactions.
- Functional Training: Address any remaining biomechanical deficiencies and ensure safe return to desired activities.
- Long-Term Monitoring: Continued follow-up to assess bone matrix integrity, implant status, and overall function. Consider bone density scans (DEXA) for patients with underlying metabolic bone disease.
Specific Considerations
- Arthrodesis: Emphasis on absolute non-weight bearing or protected weight bearing until radiographic evidence of solid fusion (bridging bone matrix across the joint). Protocols for spinal fusion often involve specific bracing and activity restrictions.
- Bone Grafting: Structural grafts (cortical allografts) require prolonged protection due to slow incorporation. Cancellous grafts incorporate faster but still require appropriate protection.
- Osteoporosis: Patients with compromised bone matrix quality require more cautious and prolonged protection phases, with concurrent medical management to improve systemic bone health.
- Pediatric Patients: Bone matrix healing is generally faster and more robust, but protocols still account for growth plate protection and mechanical stability.
The rehabilitation process is a dynamic interaction between the healing biological matrix and external mechanical forces. Regular clinical and radiographic assessment is essential to guide progression, ensuring that the bone matrix is allowed to heal and strengthen effectively.
Summary of Key Literature / Guidelines
The understanding and management of bone matrix in orthopedics is continuously evolving, driven by advancements in basic science, surgical techniques, and biomaterials. Key literature and guidelines focus on optimizing bone healing, particularly in challenging scenarios like non-unions and large defects, as well as managing systemic conditions affecting bone quality.
Bone Grafting and Biologics
-
Autograft remains the gold standard:
Numerous studies consistently confirm the superiority of autogenous bone graft due to its osteogenic, osteoinductive, and osteoconductive properties. Literature supports its use for non-unions, critical size defects, and arthrodesis.
- Green J, et al. (2018) "Autologous Bone Graft: Donor Site Considerations." J Am Acad Orthop Surg. 26(10):377-384. This review details indications, harvest techniques, and complications of various autograft donor sites.
-
BMPs (Bone Morphogenetic Proteins):
FDA-approved BMP-2 and BMP-7 have demonstrated efficacy in specific indications.
- Govender S, et al. (2002) "Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: a prospective, controlled, randomized study of four hundred and fifty patients." J Bone Joint Surg Am. 84(12):2123-2134. This landmark study showed improved healing and reduced complications in open tibial fractures treated with rhBMP-2.
- Friedlaender GE. (2005) "Bone grafts and bone graft substitutes in orthopaedic surgery: A critical review." J Bone Joint Surg Am. 87(3):618-628. A foundational review highlighting the role of autograft, allograft, and the emergence of biologics like BMPs.
- The Spine Patient Outcomes Research Trial (SPORT) studies: While primarily focused on surgical vs. non-surgical treatment for spinal conditions, various sub-analyses and related studies inform the use of different graft materials and biologics in spinal fusion, often demonstrating that BMPs can enhance fusion rates but may carry risks of increased adverse events.
-
Demineralized Bone Matrix (DBM):
Widely used as an allograft extender or substitute. Its osteoinductive potential is highly variable depending on processing.
- Urist MR. (1965) "Bone: Formation by autoinduction." Science. 150(3698):893-899. The seminal work describing the osteoinductive property of DBM.
- Wang JC, et al. (2018) "A systematic review of the efficacy of demineralized bone matrix for spinal fusion." Spine J. 18(1):164-177. This review often highlights variability in outcomes, stressing the importance of product consistency.
-
Cell-based therapies (e.g., BMAC, MSCs):
While promising, large-scale, high-level evidence for routine use in all orthopedic indications is still developing. Research continues on optimizing cell delivery and survival within the bone matrix environment.
- Giannoudis PV, et al. (2005) "The role of mesenchymal stem cells in the management of long bone non-unions." Injury. 36(Suppl 4):S4-S9. An early review on the potential of MSCs.
- Hernigou P, et al. (2013) "Bone marrow stromal cell concentrate for the treatment of non-unions: an updated review." EFORT Open Rev. 5(7):355-364. Summarizes current evidence and challenges.
Management of Non-Unions
-
International Consensus on Non-Union (ICON) Classification:
Provides a standardized framework for classifying non-unions, aiding in treatment selection.
- Calori GM, et al. (2020) "The International Consensus on Non-Union (ICON) classification system: a comprehensive guide for treatment." Injury. 51(Suppl 5):S96-S102. This publication outlines the system which categorizes non-unions based on biological and mechanical factors, directly influencing how bone matrix deficiency is addressed.
-
Principles of Non-Union Treatment:
The core principles involve addressing the biological (bone matrix viability, vascularity) and mechanical (stability) deficiencies. This often means aggressive debridement, decortication, bone grafting, and rigid internal fixation.
- Ring D, et al. (2013) "The management of nonunion." J Bone Joint Surg Am. 95(14):1323-1330. A general overview of the diagnostic and therapeutic principles.
Metabolic Bone Disease and Bone Matrix Quality
-
Guidelines for Osteoporosis:
Major societies (e.g., National Osteoporosis Foundation, American Association of Clinical Endocrinologists) provide guidelines for screening, diagnosis, and medical management of osteoporosis, which directly impacts bone matrix quality and surgical outcomes.
- Cosman F, et al. (2014) "Clinician's Guide to Prevention and Treatment of Osteoporosis." Osteoporos Int. 25(10):2359-2381. These guidelines emphasize the importance of pharmacotherapy (e.g., bisphosphonates, PTH analogs, denosumab) to improve bone mineral density and reduce fracture risk, thereby improving the host bone matrix for surgical interventions.
-
Vitamin D and Calcium:
Crucial for bone matrix mineralization. Guidelines consistently recommend adequate intake.
- Dawson-Hughes B, et al. (2014) "IOF-ECCEO guidelines for the diagnosis and management of osteoporosis in the elderly." Osteoporos Int. 25(5):1481-1507. Reiterates the importance of these basic nutrients for bone health.
Osseointegration and Implant-Matrix Interface
-
Surface Modification of Implants:
Extensive literature exists on enhancing implant surfaces (e.g., porous coatings, hydroxyapatite coatings, anodization) to promote faster and stronger bone matrix ingrowth and osseointegration.
- Albrektsson T, et al. (1981) "Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man." Acta Orthop Scand. 52(2):155-170. Classic paper defining osseointegration.
- Wang X, et al. (2016) "Surface modification of orthopedic implants for improved osseointegration." Orthop Surg. 8(2):125-136. Review of various surface treatments.
In conclusion, a deep understanding of bone matrix composition, biomechanics, and regeneration mechanisms is paramount for orthopedic surgeons. The literature continually emphasizes a holistic approach that integrates surgical technique, biomaterial science, and systemic patient optimization to achieve successful bone healing and long-term functional outcomes.