Articular Cartilage Damage: What You Need to Know for Healing
Introduction & Epidemiology
Articular cartilage, a highly specialized connective tissue, serves a critical role in joint function by providing a low-friction, load-bearing surface. Unlike most tissues, cartilage is avascular, aneural, and alymphatic , rendering it a tissue with a very limited intrinsic healing response . Once damaged, it lacks the capacity for significant self-repair, often leading to progressive degeneration and the eventual development of osteoarthritis. This inherent limitation in healing is a cornerstone of our understanding and approach to cartilage pathology.
The etiology of chondral lesions is multifactorial.
Chondrocyte viability is disrupted by:
*
High-impact loads:
Acute trauma, direct lacerations, or shear injuries can directly damage the chondral matrix and cellular components.
*
Prolonged excessive stress:
Chronic biomechanical overload, as seen in obesity, joint dysplasia (e.g., patellofemoral, hip), and malalignment syndromes (e.g., varus or valgus deformities), leads to sustained high stresses on chondrocytes, disrupting their metabolic function and integrity.
*
Prolonged lack of stress:
Paradoxically, inactivity or disuse, such as following joint immobilization, can also lead to cartilage atrophy and degeneration due to impaired nutrient exchange and altered biomechanical signaling pathways necessary for cartilage homeostasis.
*
Chemical issues:
The chondral environment is tightly regulated. Disruptions include:
*
Changes in pH:
Normal cartilage maintains a pH around 7.4. Inflammatory processes or metabolic derangements can lower pH, impairing chondrocyte function and accelerating matrix degradation.
*
Enzymes:
Elevated levels of matrix metalloproteases (MMPs) and aggrecanases (ADAMTS enzymes), often stimulated by inflammatory cytokines (e.g., IL-1β, TNF-α), actively degrade the extracellular matrix (ECM) components, particularly proteoglycans and collagen type II.
Epidemiologically, articular cartilage lesions are prevalent, affecting millions annually. The knee is the most common site, with chondral damage identified in a significant percentage of arthroscopic procedures, ranging from 60-70%. While smaller lesions may initially be asymptomatic, they represent a significant risk factor for progressive degeneration, pain, and functional impairment, especially in younger, active populations.
Surgical Anatomy & Biomechanics
Hyaline articular cartilage is a remarkably structured tissue, engineered for specific biomechanical functions. It is composed of a sparse population of chondrocytes (less than 10% of total tissue volume) embedded within an abundant extracellular matrix. The ECM primarily consists of water (65-80% of wet weight), collagen (predominantly type II, 10-20%), and proteoglycans (5-10%), with minor contributions from other proteins and glycoproteins.
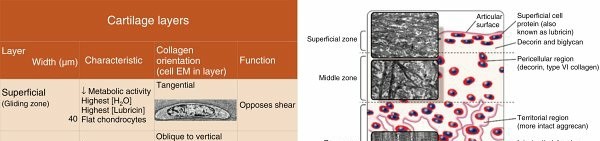
The structural organization of articular cartilage is zonal, dictating its biomechanical response:
*
Superficial (Tangential) Zone:
Characterized by flattened chondrocytes and collagen fibers oriented parallel to the articular surface. This zone resists shear forces and provides a smooth gliding surface.
*
Middle (Transitional) Zone:
Contains rounder chondrocytes and collagen fibers arranged more obliquely. It acts as a transitional layer for stress distribution.
*
Deep (Radial) Zone:
Chondrocytes are arranged in columns, and collagen fibers are oriented perpendicularly to the articular surface, anchoring the cartilage to the subchondral bone. This zone is critical for resisting compressive loads.
*
Calcified Cartilage Zone:
Separates the deep zone from the subchondral bone. It contains a higher concentration of calcium salts and is demarcated by the
tidemark
, a basophilic line that represents the interface between non-calcified and calcified cartilage. The tidemark is a crucial anatomical landmark for understanding lesion depth and healing potential.

Laceration depth is a key factor
in determining the intrinsic healing response and informing treatment strategies:
*
Lacerations above the tidemark (partial-thickness lesions):
These involve only the articular cartilage layers and do not breach the subchondral bone. They demonstrate a limited intrinsic healing potential, primarily characterized by local chondrocyte cloning, leading to limited increases in chondrocyte numbers. However, this response is often insufficient to restore the structural and functional integrity of the original hyaline cartilage, resulting in limited and often inadequate repair.
*
Lacerations extending below the tidemark into the subchondral bone (full-thickness lesions):
These lesions breach the vascularized subchondral bone, initiating an inflammatory response. This vascular access allows for the influx of blood, fibrin clot formation, and recruitment of undifferentiated mesenchymal stem cells (MSCs) from the bone marrow.
Marrow mesenchymal stem cells respond and produce less durable fibrocartilage (type I collagen)
as a repair tissue. While this fills the defect, its biomechanical properties are inferior to native hyaline cartilage, predisposing it to earlier wear and degeneration.
Blunt trauma and strenuous loading can directly lead to chondrocyte apoptosis and necrosis. The initial effects manifest as cartilage thinning and proteoglycan loss from the ECM, processes that phenotypically resemble the early stages of osteoarthritis. Furthermore, joint immobilization leads to atrophy or cartilage degeneration by altering the mechanotransductive signals necessary for chondrocyte health and impairing nutrient diffusion within the avascular matrix.
Indications & Contraindications
The decision-making process for intervention in articular cartilage damage is complex, involving consideration of patient factors, lesion characteristics, and available treatment modalities. The ICRS (International Cartilage Repair Society) grading system provides a standardized method for classifying lesion severity, which directly informs management.
- Grade 0: Normal cartilage.
- Grade 1: Nearly normal, superficial lesions only. These involve surface irregularities, softening, or fibrillation without full-thickness loss.
- Grade 2: Abnormal, lesions extend <50% of cartilage depth.
- Grade 3: Severely abnormal, lesions extend >50% of cartilage depth but do not breach the subchondral bone.
- Grade 4: Severely abnormal, lesions extend through the subchondral bone (full-thickness defects). This grade represents the primary indication for most reparative and restorative surgical procedures.
General Indications for Operative Intervention:
* Symptomatic full-thickness (ICRS Grade 3-4) chondral lesions, particularly in younger, active patients.
* Localized lesions (typically < 10-12 cm²), although larger lesions may be considered for allograft or advanced cell-based therapies.
* Failure of conservative management (e.g., activity modification, NSAIDs, physical therapy, injections).
* Presence of mechanical symptoms (locking, catching, giving way).
* Concomitant pathology that can be addressed simultaneously (e.g., meniscal tears, ligamentous instability, malalignment).
General Contraindications for Operative Intervention:
* Osteoarthritis (generalized degenerative changes, Kissing lesions).
* Uncorrected significant malalignment or instability.
* Active infection.
* Diffuse chondral damage (e.g., three-compartment OA).
* Inflammatory arthropathies (e.g., rheumatoid arthritis).
* Severe obesity (BMI >30-35), depending on the procedure.
* Unrealistic patient expectations or non-compliance with rehabilitation.
* Significant comorbidities precluding surgery.
* Older age (relative contraindication, dependent on physiological age and activity level).
The choice of specific surgical technique depends further on lesion size, location, and surgeon experience, which will be discussed in the "Detailed Surgical Approach / Technique" section.
Operative vs. Non-Operative Indications
| Feature | Non-Operative Management | Operative Management |
|---|---|---|
| Lesion Type | ICRS Grade 0-2 (superficial or partial-thickness defects) | ICRS Grade 3-4 (full-thickness defects) |
| Diffuse osteoarthritic changes (Kissing lesions) | Localized chondral defects (monocompartmental) | |
| Symptoms | Mild, intermittent pain; no mechanical symptoms | Persistent pain, mechanical symptoms (locking, catching) |
| Patient Age/Activity | Older, low-demand patients; individuals unwilling to undergo surgery/rehab | Younger, active patients; high-demand individuals |
| Concomitant Pathology | No significant instability, malalignment, or meniscal pathology | Correctable instability, malalignment, or meniscal pathology |
| Response to Conservative Care | Symptoms improve with physical therapy, activity modification, injections | Failure of 3-6 months of adequate conservative management |
| Comorbidities | Significant medical comorbidities precluding surgery | Medically fit for surgery and rehabilitation |

Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning is paramount for successful cartilage repair. This involves a comprehensive patient evaluation, detailed imaging, and meticulous surgical strategy.
Pre-Operative Assessment:
- History & Physical Examination: Detailed symptom analysis (pain, mechanical symptoms, onset, duration, aggravating/alleviating factors), functional assessment, and identification of concomitant pathologies (ligamentous laxity, meniscal tears, malalignment).
-
Imaging:
- Plain Radiographs: Weight-bearing anteroposterior, lateral, and Merchant/skyline views of the affected joint are essential to assess joint space narrowing, osteophyte formation, and alignment (varus/valgus, patellar tilt). Standing full-length alignment films are crucial if malalignment is suspected.
- Magnetic Resonance Imaging (MRI): High-resolution MRI (e.g., 3T with cartilage-specific sequences like fat-suppressed proton density, T2 mapping, dGEMRIC) is the gold standard for non-invasive assessment of cartilage lesion size, depth, quality of surrounding cartilage, and detection of subchondral bone edema or cysts. It also helps identify meniscal tears, ligamentous injuries, and osteochondral pathology.
- Computed Tomography (CT): Less utilized for cartilage directly, but valuable for complex osteochondral defects or for pre-operative planning of osteotomies, especially for rotational deformities.
- Pre-operative Arthroscopy: In some cases, a diagnostic arthroscopy may be performed prior to definitive open repair to confirm lesion characteristics, identify unexpected concomitant pathology, and debride unstable cartilage prior to a cell-based procedure.
Surgical Planning:
- Lesion Characterization: Precise mapping of lesion size (length x width) and depth (partial vs. full-thickness, presence of subchondral bone involvement).
- Technique Selection: Based on lesion size, location, patient age, activity level, and presence of concomitant pathology. Options range from marrow stimulation (microfracture) for smaller lesions, osteochondral grafting (autograft/allograft) for larger or osteochondral defects, to cell-based therapies (ACI/MACI) for larger chondral defects.
- Concomitant Procedures: Planning to address any malalignment (e.g., high tibial osteotomy, distal femoral osteotomy), meniscal pathology (repair, meniscectomy, transplantation), or ligamentous instability (ACL reconstruction) in a staged or concurrent manner. These are often critical for the long-term success of cartilage repair.
- Implant Selection: Appropriate sizing of osteochondral plugs, choice of membrane/scaffold for ACI/MACI, or instruments for microfracture.
Patient Positioning:
- General Considerations: The patient is typically positioned supine on a radiolucent operating table. Depending on the joint, specific techniques are employed.
- Knee: A high thigh tourniquet is applied. The limb is typically placed in a leg holder or over a lateral post, allowing for controlled flexion and extension. For open procedures, the knee may be flexed to 90 degrees or more.
- Ankle: Often positioned supine with a bump under the ipsilateral hip for internal rotation, or in a lateral decubitus position. A tourniquet is usually applied.
- Hip: Positioning depends on the approach. Anterior approach often requires supine, posterior requires lateral decubitus. Traction may be used for arthroscopic access.
- Shoulder: Beach chair or lateral decubitus position, with attention to axillary roll and nerve protection.
Appropriate padding and care must be taken to prevent iatrogenic nerve compression or skin breakdown. Prophylactic antibiotics are administered.
Detailed Surgical Approach / Technique
The landscape of articular cartilage repair is diverse, evolving from simple marrow stimulation to complex cell-based therapies. The underlying principle across most techniques is to either stimulate a repair response or replace the damaged tissue.
1. Marrow Stimulation Techniques (e.g., Microfracture)
- Mechanism: These techniques aim to create channels into the subchondral bone, allowing bone marrow elements, including mesenchymal stem cells (MSCs), growth factors, and progenitor cells, to access the chondral defect. This influx forms a fibrin clot, which then undergoes metaplasia into fibrocartilaginous repair tissue.
- Indications: Typically for smaller, contained full-thickness chondral lesions (< 2-4 cm²), especially in lower-demand patients or as a first-line treatment.
-
Surgical Steps (Arthroscopic):
- Arthroscopic Portal Placement: Standard anteromedial and anterolateral portals for visualization and instrumentation. Additional portals may be used.
- Lesion Debridement: All unstable and damaged cartilage is debrided back to a stable margin of healthy surrounding articular cartilage using curettes, shavers, or thermal probes. The lesion should be circular or oval, with a perpendicular wall of healthy cartilage.
- Calcified Cartilage Removal: The calcified cartilage layer at the base of the defect must be carefully removed to expose the subchondral bone plate. This is often done with a curette.
- Subchondral Bone Penetration: Using a specialized microfracture awl or K-wire (typically 3-4 mm apart), multiple perforations are created in the subchondral bone plate. The depth should be sufficient to observe fat droplets and blood efflux (the "champagne effect"), indicating marrow access, but care must be taken not to penetrate too deeply to avoid subchondral bone collapse or cyst formation.
- Post-Procedure: The joint is thoroughly irrigated.
- Outcomes: While effective in filling defects and providing pain relief, the repair tissue is predominantly fibrocartilage (Type I collagen), which is biomechanically inferior to native hyaline cartilage (Type II collagen). Long-term outcomes can be variable, with potential for repair tissue breakdown over time.
2. Osteochondral Autograft Transplantation (OATS / Mosaicplasty)
- Mechanism: Involves transferring one or more cylindrical osteochondral plugs from a less load-bearing donor site (e.g., intercondylar notch, superolateral femoral condyle) to the chondral defect. This replaces the damaged tissue with hyaline cartilage and subchondral bone.
- Indications: Full-thickness chondral or osteochondral defects, typically 1-10 cm² in size, especially in younger, active patients.
-
Surgical Steps (Arthroscopic or Mini-Open):
- Lesion Preparation: The recipient site is debrided to stable cartilage margins. A specialized coring reamer of appropriate diameter is used to create a defect perpendicular to the articular surface, ensuring a snug fit for the graft.
- Donor Site Selection: Non-weight-bearing areas are identified.
- Graft Harvesting: Using a matching coring reamer, one or multiple osteochondral plugs are harvested from the donor site, ensuring the articular cartilage surface is oriented correctly and the underlying bone plug is adequate.
- Graft Implantation: The osteochondral plugs are carefully inserted into the recipient site, ensuring the articular surface is flush with the surrounding native cartilage. Impaction is performed with a tamp to achieve stable press-fit fixation. Multiple smaller plugs are used in mosaicplasty to fill larger defects.
- Outcomes: Produces hyaline or hyaline-like repair tissue. Challenges include donor site morbidity (pain, stiffness, potential for OA), technical demands, and potential for incongruity between graft and host cartilage.
3. Osteochondral Allograft Transplantation (OCA)
- Mechanism: Similar to OATS, but uses a fresh-frozen or cryopreserved osteochondral graft from a cadaveric donor.
- Indications: Larger full-thickness chondral or osteochondral defects (> 8-10 cm²), often when autograft options are insufficient or as a salvage procedure.
-
Surgical Steps (Open):
- Graft Sizing: Pre-operative imaging and intraoperative templating are used to match the size and contour of the allograft to the recipient defect.
- Recipient Site Preparation: The defect is meticulously debrided. A specialized reamer (often cannulated for precise alignment) is used to prepare the recipient bed to match the allograft geometry.
- Allograft Preparation: The allograft is prepared (e.g., warmed, trimmed) and carefully inserted into the recipient site.
- Fixation: Press-fit is often sufficient, but supplementary fixation with bioabsorbable screws or K-wires may be used, particularly for larger grafts or less stable fixation.
- Outcomes: Can replace large defects with viable hyaline cartilage and bone. Advantages include no donor site morbidity and ability to treat larger lesions. Disadvantages include graft availability, potential for disease transmission (though rare), cost, and risk of immune response/rejection (though immune privilege of cartilage often mitigates this).
4. Autologous Chondrocyte Implantation (ACI) / Matrix-Associated Autologous Chondrocyte Implantation (MACI)
-
Mechanism:
A two-stage procedure (ACI) or a one-stage procedure (MACI) where autologous chondrocytes are harvested, expanded ex vivo, and then reimplanted into the chondral defect.
- ACI (First Generation): Chondrocytes are injected under a periosteal flap or collagen membrane sutured over the defect.
- MACI (Second/Third Generation): Chondrocytes are cultured onto a scaffold (e.g., collagen membrane, hyaluronic acid matrix) in vitro, and then this construct is implanted into the defect. This simplifies the surgical technique, avoids the need for a periosteal harvest, and provides a stable cellular construct.
- Indications: Symptomatic full-thickness chondral defects, typically 2-12 cm², in younger patients (under 50-55 years old) without advanced OA.
-
Surgical Steps (Open or Arthroscopic-Assisted):
- Stage 1 (Harvest): A small (200-300 mg) cartilage biopsy is harvested from a non-weight-bearing area (e.g., intercondylar notch) via arthroscopy. The biopsy is sent to a specialized lab for chondrocyte isolation and expansion. This process takes 3-6 weeks.
-
Stage 2 (Implantation):
- Open Arthroplasty: The joint is opened, the defect is debrided to stable perpendicular walls down to the subchondral bone, removing the calcified cartilage layer.
- Lesion Measurement & Template: The defect is carefully measured, and a template (for ACI) or the scaffold (for MACI) is sized.
- ACI: A periosteal flap (harvested from the medial tibia) or a collagen membrane is trimmed to size and sutured meticulously to the surrounding cartilage, creating a watertight compartment over the defect. The expanded chondrocytes (in suspension) are then injected under this cover.
- MACI: The cell-seeded membrane/scaffold is trimmed to precisely fit the debrided defect. It is then placed into the defect with the cellular side facing inwards (towards the bone) or outwards (towards the joint surface, depending on the specific product) and secured with fibrin glue, ensuring full coverage and stable fixation.
- Outcomes: ACI/MACI typically produces a hyaline-like repair tissue (Type II collagen predominant) with better long-term durability than fibrocartilage. However, it is a demanding procedure with a prolonged rehabilitation, and potential complications include graft hypertrophy (ACI), delamination, or incomplete fill.
5. Scaffold-Based and Newer Techniques
-
Mechanism:
Utilizing various synthetic or biologic scaffolds (e.g., collagen, hyaluronic acid, synthetic polymers) designed to provide a template for cellular ingrowth and differentiation. These can be used with or without exogenously added cells or growth factors.
- Autologous Matrix-Induced Chondrogenesis (AMIC): Combines microfracture with a resorbable collagen membrane placed over the microfractured bed to stabilize the fibrin clot and guide MSC differentiation.
- Biologic Augmentation: Addition of platelet-rich plasma (PRP), bone marrow aspirate concentrate (BMAC), or other growth factors to enhance the repair response of any chosen technique.
- Stem Cell-Based Therapies: Direct implantation of allogeneic or autologous MSCs (e.g., from adipose tissue or synovial fluid) into defects, sometimes combined with scaffolds. These are still largely experimental or in advanced clinical trials.
- Indications: Variable, ranging from small to medium-sized chondral defects, often as an alternative to ACI/MACI or to augment microfracture.
- Surgical Steps: Technique varies widely based on the specific scaffold and whether it's combined with microfracture or cell implantation. Generally involves debriding the defect, preparing the subchondral bone (e.g., microfracture), and then securing the scaffold with fibrin glue or sutures.
- Outcomes: Promising early results, with ongoing research to optimize scaffold properties, cell delivery, and growth factor application. Aims to achieve hyaline cartilage regeneration more consistently.
Concomitant Procedures
No cartilage repair procedure exists in a vacuum. It is critical to address any underlying biomechanical abnormalities or associated soft tissue injuries to ensure the long-term success of the cartilage repair.
*
Alignment Correction:
Osteotomies (e.g., High Tibial Osteotomy for medial compartment lesions with varus alignment, Distal Femoral Osteotomy for lateral compartment lesions with valgus alignment) are frequently performed, either concurrently or as a staged procedure, to offload the repaired cartilage.
*
Meniscal Pathology:
Meniscal tears (repair or partial meniscectomy) or meniscal transplantation are often necessary to restore joint stability and load distribution.
*
Ligamentous Instability:
Ligament reconstruction (e.g., ACL reconstruction) is vital to prevent abnormal shear forces and protect the repaired cartilage.

Complications & Management
Despite advances in technique, cartilage repair procedures are associated with potential complications, which can impact functional outcomes. Understanding and proactively managing these risks is crucial.
| Complication | Incidence (%) | Salvage Strategies |
|---|---|---|
| Graft Failure/Delamination | 5-20% (varies by technique) | Revision with same or alternative cartilage repair technique, osteochondral allograft, arthroplasty (for severe failure) |
| Hypertrophy (ACI) | 10-30% | Arthroscopic debridement/shaving, possibly revision with MACI or other technique |
| Incomplete Fill/Non-integration | 5-15% (OATS/OCA), 5-10% (ACI/MACI) | Microfracture augmentation, targeted debridement and application of biologic adjuncts, revision grafting |
| Donor Site Morbidity (OATS) | 5-10% | NSAIDs, physical therapy, occasional debridement for pain/cysts; rarely, revision grafting to donor site |
| Arthrofibrosis/Stiffness | 5-20% | Aggressive physical therapy, manipulation under anesthesia, arthroscopic lysis of adhesions |
| Infection | <1% (rare) | Irrigation & debridement, targeted antibiotics, hardware removal if present |
| Pain/Swelling | Common post-op, persistent can be 5-10% | NSAIDs, physical therapy, steroid injections, workup for underlying causes (e.g., synovitis, loose bodies) |
| Progression of OA | Long-term risk, ~10-20% at 5-10 years | Symptomatic management, repeat cartilage procedures, eventually arthroplasty |
| Nerve/Vascular Injury | <1% (rare) | Early recognition and repair, supportive care |
| Fracture of Host Bone/Graft | <1% (rare, OATS/OCA) | Surgical fixation, bone grafting, revision with larger or alternative graft |
Management Principles:
- Early Recognition: Vigilant post-operative monitoring and prompt investigation of new or worsening symptoms are critical.
- Conservative Measures First: Many complications, especially stiffness or persistent pain, can initially be managed with physical therapy, activity modification, anti-inflammatory medications, and targeted injections.
- Imaging: Repeat MRI is often invaluable for diagnosing graft delamination, hypertrophy, or subchondral bone issues.
- Arthroscopic Debridement: For issues like graft hypertrophy or mechanical impingement from unstable repair tissue.
- Revision Surgery: For significant graft failure, non-integration, or persistent symptoms, revision with the same or an alternative cartilage repair technique, or potentially conversion to an arthroplasty, may be necessary.
- Address Underlying Issues: If malalignment or instability was not adequately addressed initially, this must be considered in the context of revision surgery.
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is as crucial as the surgical procedure itself for the success of cartilage repair. The goal is to protect the healing tissue while gradually restoring range of motion, strength, and function. Protocols vary significantly based on the surgical technique, lesion size, and location, but generally follow a phased approach. Joint immobilization leads to atrophy or cartilage degeneration , thus controlled motion is essential, balanced with protective measures.
General Principles:
- Protection: Shielding the newly repaired or regenerated tissue from excessive load or shear forces.
- Controlled Motion: Early, protected range of motion (ROM) to promote nutrient diffusion, reduce stiffness, and encourage proper collagen alignment.
- Progressive Loading: Gradual increase in weight-bearing and impact activities.
- Neuromuscular Re-education: Restoring proprioception, balance, and coordination.
- Strength Training: Strengthening surrounding musculature to improve joint stability and shock absorption.
Phase 1: Protection & Early Motion (Weeks 0-6/8)
- Goal: Protect repair site, minimize pain/swelling, restore initial ROM.
-
Weight-Bearing (WB):
- Microfracture/ACI/MACI: Non-weight-bearing (NWB) or touch-down weight-bearing (TDWB) for 4-6 weeks, especially for femoral condyle lesions. Some protocols allow partial weight-bearing (PWB) earlier for patellofemoral lesions.
- OATS/OCA: NWB/TDWB for 4-8 weeks to allow for graft incorporation, longer for larger grafts or those requiring osteotomy.
- Immobilization: Often a brace is used for protection, sometimes locked in extension for NWB, unlocked for ROM exercises.
-
Range of Motion (ROM):
- Continuous Passive Motion (CPM): Often initiated immediately post-op (4-6 hours daily for 4-6 weeks) to enhance nutrient diffusion and prevent adhesions.
- Passive & Assisted ROM: Within pain-free limits.
- Therapeutic Exercises: Isometric exercises (quad sets, glute sets), ankle pumps. Upper body and core strengthening.
- Edema/Pain Management: Ice, elevation, compression.
Phase 2: Gradual Loading & Strengthening (Weeks 6/8-12/16)
- Goal: Increase weight-bearing, progress ROM, initiate low-impact strengthening.
- Weight-Bearing: Progress from TDWB/PWB to full weight-bearing (FWB) over several weeks, often guided by clinical exam and pain tolerance.
- ROM: Continue to increase, aiming for full pain-free ROM.
-
Strengthening:
- Closed-Chain Exercises: Wall slides, mini-squats (shallow), leg press (controlled range).
- Open-Chain Exercises: Light hamstring curls, knee extensions (avoiding excessive loading in critical ranges for patellofemoral lesions).
- Proprioception: Balance exercises.
- Modalities: May continue with modalities as needed for pain/swelling.
- Aqua Therapy: Excellent for initiating weight-bearing and functional movements with reduced joint stress.
Phase 3: Advanced Strengthening & Return to Activity (Months 4-6+)
- Goal: Restore strength, power, endurance, and prepare for sport-specific activities.
- Strengthening: Advance resistance and complexity of exercises. Lunges, step-ups, single-leg squats. Plyometrics (low-level initially).
- Functional Training: Agility drills, sport-specific movements (e.g., cutting, pivoting), gradually increasing intensity.
-
Return to Activity:
- Low-Impact Activities: Cycling, swimming, elliptical machine can typically begin around 4-6 months.
- High-Impact/Cutting Sports: Typically not recommended until 9-12 months post-op, and often later for more complex repairs or those involving osteotomies. Requires careful functional testing and surgeon clearance.
- Patient Education: Emphasis on long-term joint protection, maintaining ideal body weight, and recognizing signs of potential overload.
Specific Considerations by Technique:
- OATS/OCA: Greater emphasis on protecting the graft site during initial weight-bearing.
- ACI/MACI: The early phases are crucial for cellular integration and maturation. Activity progression may be slower, especially for high-impact activities. Hypertrophy of the repair tissue can sometimes necessitate modification of rehabilitation or even arthroscopic debridement.
- Concomitant Procedures (e.g., Osteotomy): Rehabilitation must integrate the specific protocols for the osteotomy (e.g., delayed weight-bearing for bone healing) with the cartilage repair protocol.
Adherence to the rehabilitation protocol is paramount. Deviation or premature return to high-impact activities significantly increases the risk of graft failure and recurrent symptoms.
Summary of Key Literature / Guidelines
The field of articular cartilage repair is dynamic, with ongoing research refining existing techniques and exploring novel biological solutions. Key literature and guidelines provide evidence-based frameworks for clinical practice.
1. ICRS Guidelines: The International Cartilage Repair Society (ICRS) provides consensus statements and treatment algorithms that are widely recognized. Their grading system for chondral lesions is a universal standard for characterization and prognostication. The ICRS also supports research and hosts conferences that disseminate the latest advancements.
2. Comparative Studies:
*
Microfracture vs. ACI/MACI:
Numerous studies and meta-analyses have compared marrow stimulation techniques (microfracture) with cell-based therapies (ACI/MACI). Generally, ACI/MACI tend to yield superior outcomes in terms of durability and quality of repair tissue (hyaline-like) for larger lesions, particularly in younger patients, though with higher cost and longer rehabilitation. Microfracture remains a viable and cost-effective option for smaller lesions, but its limitations regarding fibrocartilage repair are well-documented.
*
OATS vs. ACI/MACI:
For osteochondral lesions, OATS and OCA have shown excellent results, directly replacing the defect with hyaline cartilage and bone. While OATS has donor site morbidity, OCA addresses this, but with concerns about graft availability and handling. ACI/MACI are primarily for chondral defects and require a stable subchondral bone bed.
3. Biologics and Tissue Engineering:
This is a rapidly advancing frontier.
*
Growth Factors:
The addition of Platelet-Rich Plasma (PRP) or Bone Marrow Aspirate Concentrate (BMAC) to existing repair techniques (e.g., microfracture, AMIC) has shown promise in enhancing chondrogenesis and improving repair tissue quality, though large-scale, high-quality clinical trials are still ongoing to definitively establish efficacy and optimal application protocols.
*
Scaffold Design:
Research is focused on developing biodegradable scaffolds with specific architectural properties, porosity, and biomimetic cues to guide chondrocyte or MSC differentiation into durable hyaline cartilage. These include biphasic scaffolds for osteochondral defects and scaffolds incorporating growth factors or gene therapy constructs.
*
Allogeneic Stem Cells:
The use of allogeneic MSCs (from donors) or induced pluripotent stem cells (iPSCs) offers potential advantages by overcoming donor site morbidity and allowing for off-the-shelf products, though challenges related to immunogenicity and regulatory hurdles remain.
4. Alignment and Concomitant Pathology: Strong evidence supports the necessity of addressing underlying biomechanical factors. Studies consistently show that uncorrected malalignment or ligamentous instability significantly compromises the long-term success of any cartilage repair procedure. This underscores the holistic approach required for optimal patient outcomes.
5. Long-term Outcomes: While many cartilage repair techniques demonstrate good to excellent short- to mid-term outcomes (5-10 years), the long-term durability and ability to truly prevent the onset of osteoarthritis remain areas of active investigation. Patient-specific factors, meticulous surgical technique, and rigorous adherence to rehabilitation protocols are consistently identified as key determinants of long-term success. The progression to secondary osteoarthritis, despite successful initial repair, is a recognized challenge, particularly for patients with higher BMIs, greater baseline degenerative changes, or recurrent injuries.
In conclusion, the management of articular cartilage damage demands a deep understanding of its unique biological properties, a nuanced approach to surgical technique selection, and an appreciation for the critical role of comprehensive post-operative rehabilitation. As academic orthopedic surgeons and educators, we must stay abreast of the evolving evidence base to provide the most effective and durable solutions for our patients.