Spinal Nerve Anatomy & Surgical Repair: Orthopaedic Guide
Key Takeaway
The anatomy of the spinal nerves dictates both the clinical presentation of neurological deficits and the surgical strategies required for repair. Understanding the microscopic topography, vascular supply, and functional components of mixed spinal nerves is paramount. This guide details the gross and microscopic anatomy, dermatomal distributions, and evidence-based operative techniques for managing peripheral nerve injuries, including sciatic, peroneal, and tibial nerve repairs.
INTRODUCTION TO SPINAL NERVE ANATOMY
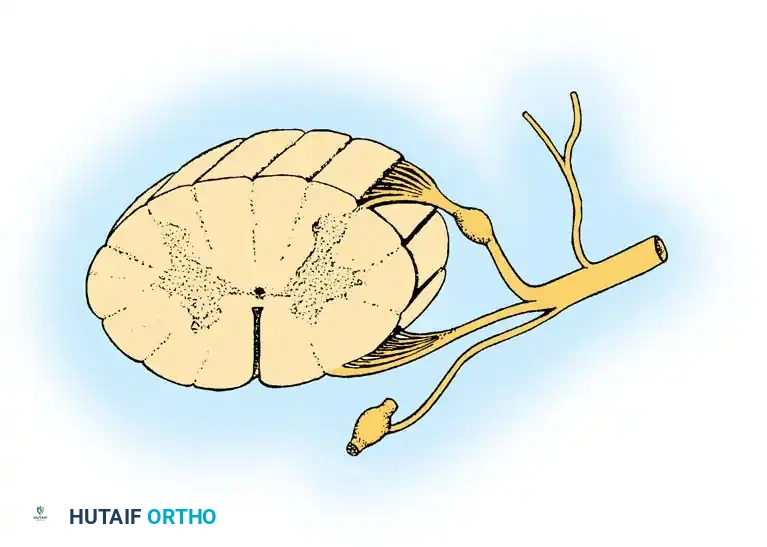
A profound understanding of spinal nerve anatomy is the cornerstone of orthopaedic neurology, spine surgery, and peripheral nerve reconstruction. Each segmental spinal nerve represents a critical junction between the central nervous system (CNS) and the peripheral nervous system (PNS). Formed at or near its respective intervertebral foramen, the spinal nerve is created by the union of its dorsal (sensory) root and its ventral (motor) root.
In the human body, a total of 31 pairs of mixed spinal nerves exit their respective foramina to innervate the homolateral trunk and extremities. These are categorized as:
* 8 Cervical (C1–C8)
* 12 Thoracic (T1–T12)
* 5 Lumbar (L1–L5)
* 5 Sacral (S1–S5)
* 1 Coccygeal (Co1)
In most thoracic segments, these mixed spinal nerves retain their primitive autonomy, supplying a single intercostal dermatomal and myotomal segment. However, in virtually all other segments of the spinal axis, the anterior primary rami join to form complex plexuses (cervical, brachial, lumbar, and lumbosacral) that innervate the limbs, abandoning the primitive myomeric pattern to accommodate the complex biomechanical demands of the extremities.
GROSS ANATOMY AND FUNCTIONAL COMPONENTS
A typical mixed spinal nerve is a highly organized conduit comprising three distinct functional components: motor, sensory, and sympathetic.

Motor Components (Somatic Efferent)
Several rootlets leave the anterolateral sulcus of the spinal cord and unite to form each ventral (motor) root. The fibers traversing these roots arise directly from the anterior horn cells of the spinal cord gray matter. These heavily myelinated alpha motor neurons are responsible for the direct innervation of skeletal muscles, dictating myotomal function.
Sensory Components (Somatic Afferent)
Sensory fibers arise from peripheral receptors detecting pain, thermal changes, tactile stimuli, and stretch (proprioception). The cell bodies for these pseudounipolar neurons are housed within the dorsal root ganglia (DRG), which typically reside within the intervertebral foramen. Axons from the DRG enter the posterolateral sulcus of the spinal cord via several dorsal rootlets.
Central projections follow distinct pathways:
* Dorsal Columns (Medial Lemniscal Pathway): Fibers conveying joint position sense (proprioception) and fine tactile discrimination turn cephalad in the dorsal columns. They ascend ipsilaterally without synapsing until they reach the gracile and cuneate nuclei at the cervicomedullary junction.
* Spinothalamic Tracts: Pain and temperature fibers synapse immediately in the substantia gelatinosa of the dorsal horn, cross the midline via the anterior white commissure, and ascend in the contralateral lateral spinothalamic tract. Crude tactile fibers synapse and cross to ascend in the ventral spinothalamic tract.
Sympathetic Components (Autonomic)
The sympathetic component of all 31 mixed spinal nerves originates from only 14 motor roots. The cells of origin are located in the intermediolateral cell column, which extends from the first thoracic (T1) to the second lumbar (L2) cord segments.
These preganglionic efferent fibers exit the cord via the T1–L2 motor roots, enter the mixed spinal nerve, and promptly emerge as white rami communicantes. They pass anteriorly to the corresponding paravertebral sympathetic ganglion. From here, fibers may synapse immediately, or travel up or down the sympathetic chain. Postganglionic fibers then return to the mixed spinal nerves via gray rami communicantes. Because gray rami are unmyelinated, they are found at all 31 spinal levels, providing sympathetic innervation (sudomotor to sweat glands, vasomotor to blood vessels, and pilomotor to erector pili muscles) in a segmental pattern.
Clinical Pearl: The presence of Horner's syndrome (ptosis, miosis, anhidrosis) in the setting of a brachial plexus injury indicates a proximal avulsion of the T1 nerve root, as the sympathetic ganglion is disrupted near its origin. This carries a poor prognosis for spontaneous motor recovery.
DERMATOMAL AND MYOTOMAL MIGRATION
Upon exiting the intervertebral foramina and receiving their sympathetic components, mixed spinal nerves promptly bifurcate into anterior and posterior primary rami.
Posterior Primary Rami
The posterior primary rami are directed dorsally to supply the paraspinal musculature (erector spinae) and the overlying skin of the posterior trunk, neck, and head. Notably, the upper three cervical posterior rami are larger than their anterior counterparts, supplying the posterior scalp and craniocervical musculature. Elsewhere, posterior rami remain small.
Anterior Primary Rami and Plexus Formation
The major portion of each spinal nerve continues laterally as the anterior primary ramus. While thoracic rami become intercostal nerves, the cervical, lower lumbar, and sacral rami form plexuses.
* Cervical Plexus: C1–C4
* Brachial Plexus: C5–T1
* Lumbar Plexus: L1–L4
* Lumbosacral Plexus: L4–S4
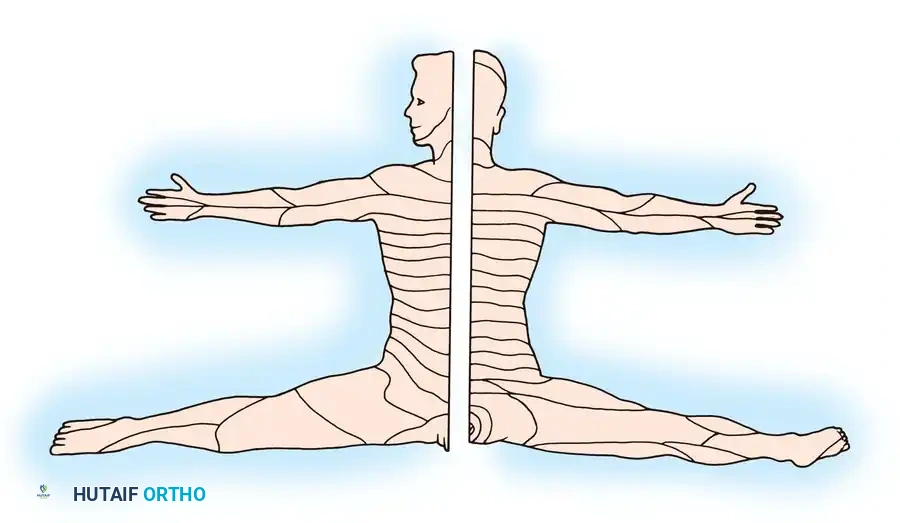
The embryological enlargement and prolongation of the limb buds markedly alter the primitive metameric pattern. Dermatomes (the area of skin supplied by a single spinal root) and myotomes migrate into the developing limbs. This migration displaces midcervical dermatomes along the lateral arm and radial forearm, while lower cervical and upper thoracic dermatomes are pulled along the medial arm and ulnar forearm. The line separating rostral from caudal segmental dermatomes is termed the axial line.
MICROSCOPIC ANATOMY AND INTRANEURAL TOPOGRAPHY
Surgical repair of peripheral nerves demands a rigorous understanding of their microscopic architecture. Each nerve fiber (axon) is a direct extension of a neuron and is either myelinated or unmyelinated. Sensory and motor nerves contain both, typically in a ratio of 4:1.
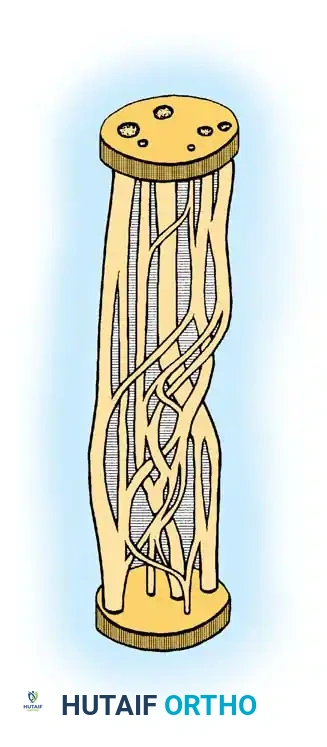
Schwann Cells and Myelination
In unmyelinated fibers, a single Schwann cell envelops several axons. In heavily myelinated fibers, a Schwann cell rotates to form a multilaminated myelin sheath around a single axon. The myelinated segment enclosed by one Schwann cell is the internode (0.1 to 1.8 mm in length). The myelin-free gap between adjacent Schwann cells is the Node of Ranvier, which facilitates rapid saltatory conduction.
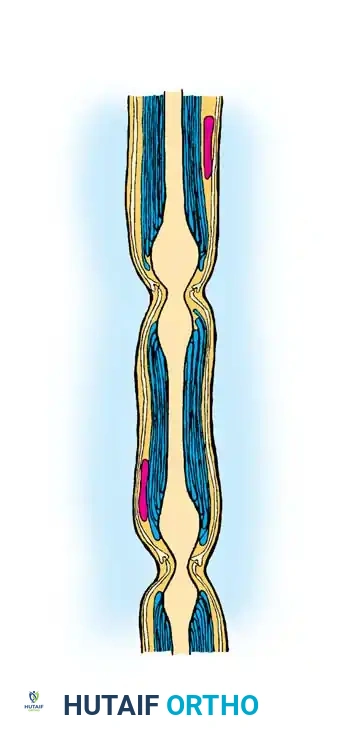
Connective Tissue Sheaths
The structural integrity of a peripheral nerve is maintained by three distinct connective tissue layers:
1. Endoneurium: A delicate veil of fibrous tissue surrounding individual axons and their Schwann cells.
2. Perineurium: A dense, metabolically active layer surrounding a group of axons to form a fascicle (or funiculus). The perineurium provides the primary tensile strength of the nerve and maintains the blood-nerve barrier.
3. Epineurium: The outermost layer of loose connective tissue encasing the entire group of fascicles, forming the macroscopic peripheral nerve. It cushions the fascicles against compressive forces.
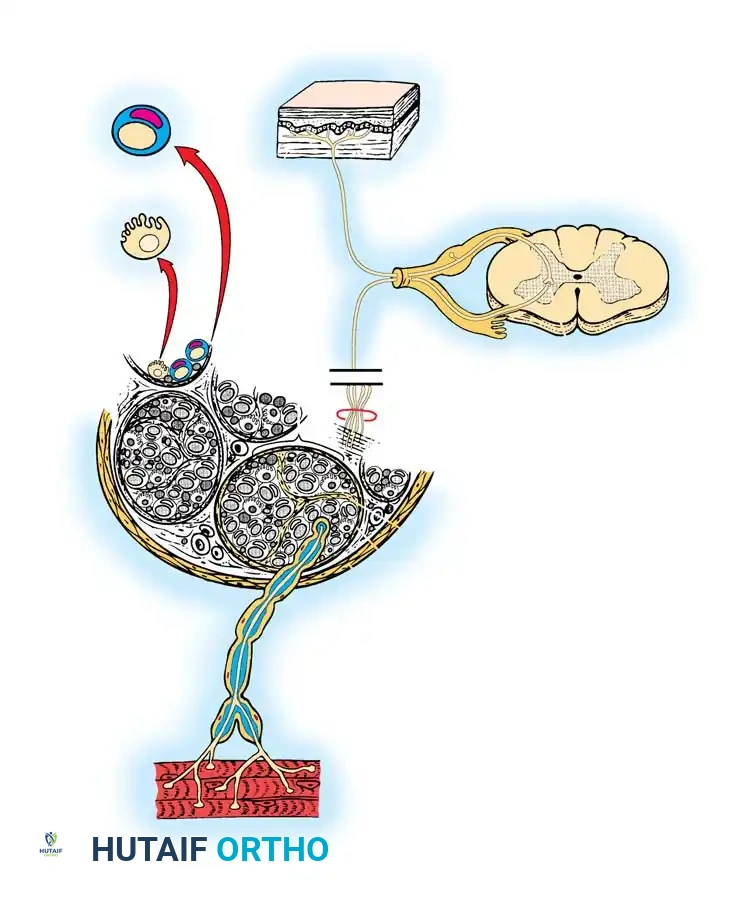
Vascular Supply (Vasa Nervorum)
The blood supply to a peripheral nerve enters through the mesoneurium, a loose connective tissue suspensory ligament extending from the epineurium to surrounding fascial beds.
* Extrinsic System: Segmental vessels entering via the mesoneurium.
* Intrinsic System: A highly anastomotic longitudinal network within the epineurium, perineurium, and endoneurium.
Surgical Warning: The extensive intrinsic longitudinal blood supply allows for surgical mobilization of nerves over variable lengths. However, over-mobilization or aggressive stripping of the epineurium can devascularize the nerve, leading to ischemic fibrosis and failure of nerve regeneration.
SURGICAL MANAGEMENT OF PERIPHERAL NERVE INJURIES
Understanding the anatomy of spinal nerves and their peripheral extensions is critical when addressing traumatic nerve injuries. The principles of nerve repair apply universally, but specific approaches are required for major lower extremity nerves.
General Principles of Nerve Repair
- Examination: Accurate preoperative clinical examination (motor grading, sensory mapping, Tinel's sign) and electrodiagnostic studies (EMG/NCS) are mandatory to localize the lesion.
- Critical Limit of Delay: Motor endplates undergo irreversible fibrosis if denervated for prolonged periods. The critical limit of delay for nerve suture is generally 12 to 18 months; however, repairs performed within the first 3 to 6 months yield significantly superior functional outcomes.
- Methods of Closing Gaps: Primary end-to-end neurorrhaphy must be tension-free. If a gap exists, options include:
- Extensive proximal and distal mobilization.
- Joint positioning (e.g., flexing the knee for sciatic/tibial repairs).
- Bone shortening (rarely indicated solely for nerve repair).
- Autologous Nerve Grafting: The gold standard for gaps that cannot be closed without tension. The sural nerve is the most common donor.
Sciatic Nerve Surgery
The sciatic nerve (L4-S3) is the largest nerve in the body, highly susceptible to injury during posterior hip dislocations, pelvic fractures, or iatrogenically during total hip arthroplasty.
- Surgical Approach: The patient is positioned prone. A curvilinear incision is made from the posterior superior iliac spine (PSIS), extending distally over the greater trochanter, and curving down the posterior midline of the thigh. The gluteus maximus is split in line with its fibers or reflected to expose the nerve as it exits the greater sciatic foramen below the piriformis.
- Results of Suture: Recovery is heavily dependent on the level of injury. Proximal lesions have a poorer prognosis for distal motor recovery (intrinsic foot muscles) due to the distance regenerating axons must travel (1 mm/day). The tibial division typically recovers better than the peroneal division due to its larger fascicular cross-sectional area and deeper anatomical protection.
Peroneal Nerve Surgery
The common peroneal nerve is highly vulnerable to compression or laceration at the fibular neck. Paralysis results in a devastating "foot drop" (loss of ankle dorsiflexion and toe extension).
- Surgical Approach: The nerve is approached via a lateral incision over the biceps femoris tendon, extending distally across the fibular neck. The nerve must be carefully decompressed as it dives beneath the fibrous arch of the peroneus longus muscle.
- Tendon Transfer for Peroneal Nerve Paralysis: If nerve repair fails or the critical limit of delay has passed, tendon transfers are highly effective. The standard procedure is the transfer of the Posterior Tibial Tendon (PTT). The PTT is harvested, routed through the interosseous membrane, and inserted into the middle cuneiform or split to the tibialis anterior and peroneus brevis to restore balanced dorsiflexion.
Tibial Nerve Surgery
The tibial nerve provides motor innervation to the posterior compartment of the leg and plantar aspect of the foot, and sensory innervation to the sole.
- Approach in the Popliteal Fossa: The patient is prone. An S-shaped incision is utilized across the popliteal crease to avoid flexion contractures. The deep fascia is incised, and the nerve is identified superficial and lateral to the popliteal vein and artery.
- Methods of Closing Gaps: Gaps in the popliteal fossa can often be overcome by generous mobilization of the nerve proximally into the thigh and distally beneath the soleus arch, combined with 90 degrees of knee flexion. Postoperatively, the knee is immobilized in flexion and gradually extended over 4 to 6 weeks to prevent traction injury to the repair site.
- Results of Suture: Tibial nerve repairs generally yield favorable motor outcomes for the gastrocnemius-soleus complex. However, return of intrinsic foot motor function and fine two-point discrimination on the plantar surface is often incomplete, necessitating vigilant postoperative foot care to prevent neuropathic ulceration.
POSTOPERATIVE PROTOCOLS
Following any peripheral nerve repair or grafting, the surgical site must be protected from tension.
1. Immobilization: The limb is splinted in a position that minimizes tension on the neurorrhaphy for 3 to 4 weeks.
2. Mobilization: Gradual extension of the joints is initiated. Aggressive stretching is strictly contraindicated.
3. Rehabilitation: Physical therapy focuses on maintaining passive range of motion in denervated joints to prevent contractures. Galvanic stimulation of denervated muscle remains controversial but is used in some centers.
4. Monitoring: Clinical progression is monitored via the advancing Tinel's sign (expected at approximately 1 mm per day or 1 inch per month) and serial EMG studies beginning at 3 months postoperatively.
Surgical Pearl: When performing an epineurial repair, meticulous alignment of the superficial epineurial vessels ensures correct rotational alignment of the internal fascicular topography, maximizing the chance of appropriate motor-to-motor and sensory-to-sensory axonal regeneration.
You Might Also Like

