Acute Compartment Syndrome of the Upper Extremity: Pathophysiology, Diagnosis, and Surgical Decompression
Key Takeaway
Acute compartment syndrome is a surgical emergency characterized by elevated intracompartmental pressure leading to irreversible myoneural ischemia. Early clinical recognition, particularly pain on passive stretch, and prompt surgical decompression via fasciotomy are paramount. This guide details the pathophysiology, diagnostic pressure thresholds, and step-by-step surgical techniques for forearm and hand fasciotomies to prevent Volkmann ischemic contracture.
INTRODUCTION AND PATHOPHYSIOLOGY
Acute compartment syndrome (ACS) of the upper extremity represents a catastrophic orthopedic emergency characterized by increased pressure within a closed osteofascial space. This elevated pressure compromises the local arteriovenous gradient, leading to diminished capillary perfusion, profound tissue hypoxia, and ultimately, irreversible myoneural necrosis. If left untreated or diagnosed late, ACS culminates in Volkmann’s ischemic contracture—a devastating condition marked by fibrotic muscle replacement, severe neurologic deficit, and a rigid, nonfunctional limb.
The fundamental pathomechanics of ACS are driven by the traumatic ischemia-edema cycle. Initial trauma, arterial occlusion, or arterial spasm triggers a vasospastic antidromic reflex. This leads to venous and lymphatic compression, which subsequently elevates intramuscular pressure. The resulting intramuscular edema exacerbates venous obstruction, increasing capillary permeability and stimulating histamine release. This vicious cycle rapidly accelerates, further driving up intracompartmental pressure until capillary perfusion ceases entirely.
Clinical Pearl: The tolerance of tissue to prolonged ischemia is highly variable and tissue-specific. Matsen et al. demonstrated that skeletal muscle exhibits functional impairment after merely 2 to 4 hours of ischemia, progressing to irreversible functional loss and necrosis after 4 to 12 hours. Conversely, peripheral nerve tissue demonstrates abnormal function (paresthesia) after just 30 minutes of ischemia, with irreversible structural and functional loss occurring between 12 and 24 hours.
ETIOLOGY
Numerous traumatic and atraumatic insults can precipitate compartment syndrome. In the upper extremity, high-energy crush injuries and fractures of the forearm or elbow are the most frequent culprits.
Specific etiologies include:
* Fractures: Supracondylar fractures of the humerus (particularly in the pediatric population) and both-bone forearm fractures carry the highest risk.
* Crush Injuries: Direct blunt trauma causing massive muscle contusion and hemorrhage.
* Prolonged External Compression: Tight casts, constrictive dressings, or prolonged limb positioning during prolonged surgical procedures or drug-induced coma.
* Reperfusion Injury: Following arterial bypass or repair of a vascular injury, the sudden restoration of blood flow to an ischemic limb causes massive capillary leakage and explosive compartment swelling.
* Iatrogenic Causes: Extravasation of intravenous fluids or pressurized contrast media.
CLINICAL DIAGNOSIS
A high index of suspicion is the surgeon's greatest asset. A crush injury or fracture of the forearm or elbow, especially in the supracondylar area of the humerus, must immediately raise the suspicion of an impending forearm compartment syndrome. Early diagnosis is absolutely essential because irreversible myoneural damage occurs rapidly.
Sheridan and Matsen reported critical outcome data regarding surgical timing: when fasciotomy was performed within 12 hours from the onset of compartment syndrome, normal limb function was regained in 68% of patients. Tragically, when fasciotomy was delayed 12 hours or more, only 8% of patients regained normal function.
Symptomatology and Physical Examination
The classic "6 Ps" (Pain, Pallor, Pulselessness, Paresthesia, Paralysis, Poikilothermia) are frequently taught, but relying on them is a dangerous clinical pitfall. Pallor, pulselessness, and paralysis are late, often irreversible signs.
- Pain Out of Proportion: Pain is the earliest and most consistent symptom. It is typically described as deep, unremitting, and poorly localized. Crucially, this symptom may be masked in comatose, polytraumatized, or regionally anesthetized patients.
- Pain on Passive Stretch: Passive extension of the fingers (stretching the ischemic flexor mass) dramatically exacerbates the pain. This is the most sensitive clinical sign of early ACS.
- Palpation: The forearm is exquisitely tender, tense, and exhibits a "wood-like" swelling. The dorsal compartment of the forearm may also be involved; consequently, palpation with the fingers over both volar and dorsal compartments is mandatory.
- Neurologic Deficits: Sensibility of the fingertips may be diminished early in the ischemic cascade. Two-point discrimination testing is highly valuable for determining nerve ischemia. Gelberman et al. reported that four of nine awake patients with compartment pressures greater than 30 mm Hg had median nerve 2-point discrimination of more than 1 cm.
- Vibratory Sensation: A strong correlation exists between diminished perception of vibration and increasing compartment pressure. Utilizing a 256 cycles/s tuning fork vibratory stimulus is a highly sensitive adjunct in the clinical evaluation of suspected ACS.
Surgical Warning: The radial pulse may or may not be palpable. The presence of a bounding distal pulse does not rule out compartment syndrome. Intracompartmental pressures rarely exceed systolic blood pressure; therefore, arterial inflow continues while capillary perfusion is halted.
Diagnosing Hand and Digit Compartment Syndrome
The diagnosis of a compartment syndrome within an individual interosseous muscle of the hand can be exceptionally difficult.
* Presentation: The hand is massively swollen and tense. The fingers are held almost rigid in a partially flexed position (intrinsic minus posture) with the wrist in neutral.
* Provocative Testing: Any passive movement of the fingers that causes metacarpophalangeal (MCP) joint extension stretches the intrinsic muscles and usually elicits excruciating pain.
* Adductor Compartment: The adductor compartment of the thumb can be isolated and tested by pulling the thumb into palmar abduction, thereby stretching the adductor pollicis muscle. The thenar muscles are rarely involved in isolation.
OBJECTIVE DIAGNOSTICS AND PRESSURE MONITORING
When clinical signs are equivocal—particularly in obtunded, intubated, or polytraumatized patients—objective measurement of intracompartmental pressure is mandatory. Pressures should be measured in all suspected compartments, especially the deep volar forearm, first dorsal interosseous, thenar, and hypothenar compartments.
Measurement Techniques
Several validated techniques exist for measuring compartment pressures:
1. Infusion Technique (Whitesides et al.): Measures the pressure necessary to overcome tissue pressure while injecting a minute quantity of saline into a closed compartment.
2. Wick Catheter Technique (Mubarak et al.): Allows direct measurement of compartment pressure without requiring saline infusion.
3. Slit Catheter Technique: A modification of the wick catheter method, preferred for the measurement of intermittent or chronic exertional compartment syndrome.
4. Handheld Manometer (Stryker Device): The most commonly utilized modern device. It provides rapid, accurate, and reproducible intracompartmental pressure readings via a side-port needle.
Pressure Thresholds for Fasciotomy
The absolute pressure threshold versus the differential pressure (Delta P) threshold remains a topic of academic debate, though Delta P is generally favored in modern practice.
* Whitesides Threshold (Delta P): Recommends fasciotomy when tissue pressure increases to within 10 to 30 mm Hg of the patient's diastolic blood pressure (Delta P < 30 mm Hg).
* Mubarak Threshold (Absolute): Recommends fasciotomy at an absolute measurement of 30 mm Hg.
* Matsen Threshold (Continuous): Utilizing continuous monitoring for up to 3 days, suggests that an absolute pressure of 45 mm Hg or more is a definitive indication for fasciotomy.
Advanced Imaging
While clinical examination and pressure monitoring are the gold standards for acute cases, advanced imaging has a limited but specific role.
* Noninvasive Hardness Testing: Steinberg and Gelberman reported on a noninvasive technique measuring "quantitative hardness" of limb compartments, which correlates well with direct intracompartmental pressures.
* MRI: Helpful in evaluating "muscle status" and demonstrating increased signal consistent with edema. It is highly useful in diagnosing chronic exertional compartment syndrome but is too time-consuming for acute emergencies.
* CT Scan: May localize sites of muscle necrosis but has no proven utility in diagnosing acute compartment syndrome.
SURGICAL MANAGEMENT: FASCIOTOMY OF THE UPPER EXTREMITY
The absolute treatment for acute compartment syndrome is emergent surgical decompression via fasciotomy. There is no role for conservative management once the diagnosis is confirmed. The goal is complete release of all restricting fascial envelopes.
Preoperative Preparation and Positioning
- Anesthesia: General anesthesia is preferred. Regional anesthesia is contraindicated as it masks postoperative pain, which is a critical indicator of inadequate release or recurrent compartment syndrome.
- Positioning: The patient is positioned supine with the affected arm extended on a radiolucent hand table.
- Tourniquet: A sterile tourniquet may be applied but should not be inflated unless catastrophic hemorrhage occurs. Inflating a tourniquet exacerbates ischemia and prevents the visual assessment of muscle viability following decompression.
Forearm Fasciotomy Approaches
The forearm contains three primary compartments: the volar (which is subdivided into superficial and deep), the dorsal, and the mobile wad. A complete decompression typically requires both a volar and a dorsal approach.

1. Volar Approach (Modified Henry)
The volar approach decompresses the superficial and deep volar compartments, as well as the carpal tunnel.
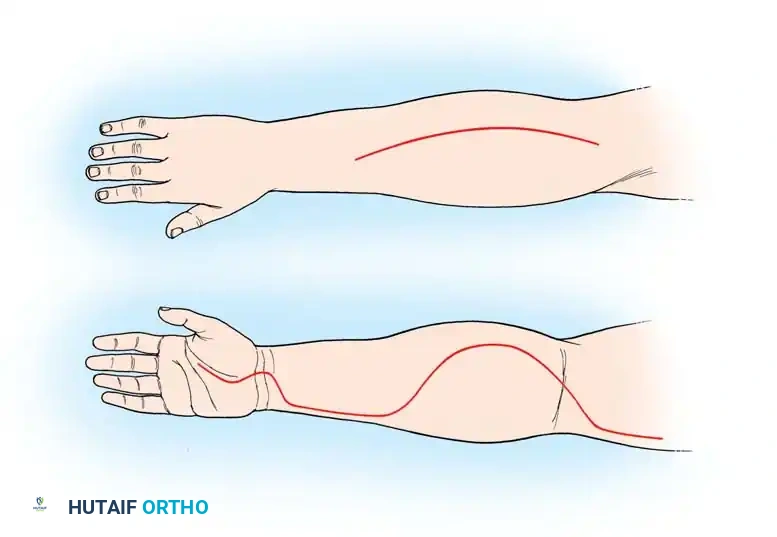
- Incision: Begin the incision proximal to the medial epicondyle. Extend it distally and laterally across the antecubital fossa to the mobile wad. Continue the incision distally along the volar forearm in a curvilinear fashion (to prevent scar contracture across the joint). As the incision approaches the wrist, curve it ulnarly to cross the wrist flexion creases obliquely, extending into the palm along the thenar crease to decompress the carpal tunnel.
- Superficial Dissection: Incise the subcutaneous tissue. Identify and protect the medial antebrachial cutaneous nerve proximally and the palmar cutaneous branch of the median nerve at the wrist.
- Fascial Release: Incise the superficial fascia of the forearm over the flexor carpi radialis (FCR).
- Deep Compartment Release: Retract the FCR ulnarly and the brachioradialis radially. This exposes the deep flexor compartment (flexor digitorum profundus and flexor pollicis longus). Incise the deep fascia longitudinally.
- Carpal Tunnel Release: Distally, divide the transverse carpal ligament completely to decompress the median nerve.
- Muscle Assessment: Inspect the muscle bellies for the "4 Cs": Color, Consistency, Contractility, and Capacity to bleed. Necrotic muscle should be meticulously debrided, though borderline muscle should be left for a second-look operation.
2. Dorsal Approach
The dorsal approach decompresses the extensor compartment and the mobile wad.
- Incision: Make a straight longitudinal incision starting from the lateral epicondyle and extending distally to the midline of the wrist.
- Fascial Release: Incise the dorsal fascia longitudinally.
- Mobile Wad: Identify the interval between the extensor digitorum communis and the mobile wad (brachioradialis, extensor carpi radialis longus, and brevis). Undermine the skin radially and incise the fascia overlying the mobile wad to ensure complete decompression.
Hand and Digit Fasciotomy
The hand contains 10 distinct compartments: four dorsal interosseous, three volar interosseous, the thenar, the hypothenar, and the adductor pollicis.
Interosseous and Adductor Compartments
- Dorsal Incisions: Make two longitudinal incisions on the dorsum of the hand. The first is placed over the second metacarpal to access the first and second dorsal interosseous compartments. The second is placed over the fourth metacarpal to access the third and fourth dorsal interosseous compartments.
- Fascial Release: Bluntly dissect through the subcutaneous tissue, protecting the dorsal sensory veins and nerves. Incise the fascia over each interosseous muscle.
- Adductor Pollicis: Through the index metacarpal incision, dissect bluntly along the ulnar border of the first metacarpal to access and release the fascia of the adductor pollicis.
Digit Fasciotomy
Digital compartment syndrome is rare but can occur in severe crush injuries. Decompression is achieved via mid-axial incisions.
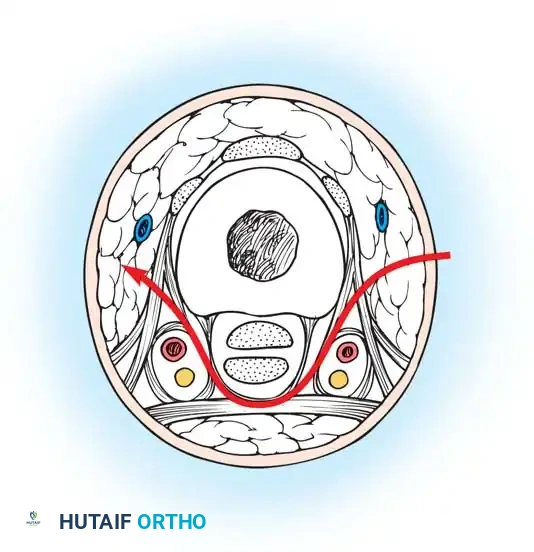
- Incision Placement: Make a mid-axial incision along the ulnar border of the index, middle, and ring fingers, and the radial border of the thumb and small finger. The incision must remain dorsal to the flexion creases.
- Dissection Path: As illustrated in the cross-section, the surgical approach must carefully navigate the digital anatomy. The incision passes through the subcutaneous fat, deliberately staying dorsal to the neurovascular bundle.
- Ligamentous Release: The dissection must pass between the Cleland ligament (dorsal) and the Grayson ligament (volar). Releasing the fascial bands here effectively decompresses the digital compartment while protecting the critical neurovascular structures.
Pitfall: Failure to accurately identify the mid-axial line can result in iatrogenic injury to the digital neurovascular bundle or cause debilitating flexion contractures if the incision crosses the volar skin creases.
POSTOPERATIVE PROTOCOL AND WOUND MANAGEMENT
Following successful fasciotomy, the surgical wounds must never be closed primarily. The massive tissue edema requires space to expand; primary closure will immediately recreate the compartment syndrome.
- Dressing: Apply a sterile, non-adherent dressing. Negative Pressure Wound Therapy (NPWT / VAC dressing) at 75 to 125 mm Hg continuous suction is highly recommended. NPWT manages exudate, reduces local edema, and prevents skin retraction.
- Immobilization: Splint the extremity in a functional position. The wrist should be extended 20 to 30 degrees, MCP joints flexed to 70 degrees, and interphalangeal joints fully extended.
- Second-Look Surgery: Return the patient to the operating room at 48 to 72 hours for a planned second look. Re-evaluate muscle viability, perform further debridement if necessary, and attempt delayed primary closure.
- Definitive Closure: If the wound edges cannot be approximated without tension by the third or fourth return to the operating room, split-thickness skin grafting is mandatory. Vessel loops woven in a "shoelace" technique can be used dynamically over several days to assist in gradual skin approximation prior to grafting.
-
Rehabilitation: Early passive and active range of motion exercises for the digits must be initiated as soon as clinically permissible to prevent tendon adhesions and joint contractures.
You Might Also Like
